A Novel Mobile App (Heali) for Disease Treatment in Participants With Irritable Bowel Syndrome: Randomized Controlled Pilot Trial
- PMID: 33650977
- PMCID: PMC7967221
- DOI: 10.2196/24134
A Novel Mobile App (Heali) for Disease Treatment in Participants With Irritable Bowel Syndrome: Randomized Controlled Pilot Trial
Abstract
Background: A diet high in fermentable, oligo-, di-, monosaccharides and polyols (FODMAPs) has been shown to exacerbate symptoms of irritable bowel syndrome (IBS). Previous literature reports significant improvement in IBS symptoms with initiation of a low FODMAP diet (LFD) and monitored reintroduction. However, dietary adherence to the LFD is difficult, with patients stating that the information given by health care providers is often generalized and nonspecific, requiring them to search for supplementary information to fit their needs.
Objective: The aim of our study was to determine whether Heali, a novel artificial intelligence dietary mobile app can improve adherence to the LFD, IBS symptom severity, and quality of life outcomes in adults with IBS or IBS-like symptoms over a 4-week period.
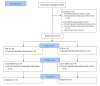
Methods: Participants were randomized into 2 groups: the control group (CON), in which participants received educational materials, and the experimental group (APP), in which participants received access to the mobile app and educational materials. Over the course of this unblinded online trial, all participants completed a battery of 5 questionnaires at baseline and at the end of the trial to document IBS symptoms, quality of life, LFD knowledge, and LFD adherence.
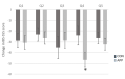
Results: We enrolled 58 participants in the study (29 in each group), and 25 participants completed the study in its entirety (11 and 14 for the CON and APP groups, respectively). Final, per-protocol analyses showed greater improvement in quality of life score for the APP group compared to the CON group (31.1 and 11.8, respectively; P=.04). Reduction in total IBS symptom severity score was 24% greater for the APP group versus the CON group. Although this did not achieve significance (-170 vs -138 respectively; P=.37), the reduction in the subscore for bowel habit dissatisfaction was 2-fold greater for the APP group than for the CON group (P=.05).
Conclusions: This initial study provides preliminary evidence that Heali may provide therapeutic benefit to its users, specifically improvements in quality of life and bowel habits. Although this study was underpowered, findings from this study warrant further research in a larger sample of participants to test the efficacy of Heali app use to improve outcomes for patients with IBS.
Trial registration: ClinicalTrials.gov NCT04256551; https://clinicaltrials.gov/ct2/show/NCT04256551.
Keywords: artificial intelligence; irritable bowel syndrome; low FODMAP diet; mobile app; randomized controlled trial.
©Aaron J Rafferty, Rick Hall, Carol S Johnston. Originally published in the Journal of Medical Internet Research (http://www.jmir.org), 02.03.2021.
Conflict of interest statement
Conflicts of Interest: None declared.
Figures
Similar articles
-
Long-Term Effects of a Web-Based Low-FODMAP Diet Versus Probiotic Treatment for Irritable Bowel Syndrome, Including Shotgun Analyses of Microbiota: Randomized, Double-Crossover Clinical Trial.J Med Internet Res. 2021 Dec 14;23(12):e30291. doi: 10.2196/30291. J Med Internet Res. 2021. PMID: 34904950 Free PMC article. Clinical Trial.
-
A low fermentable oligo-di-mono saccharides and polyols (FODMAP) diet reduced pain and improved daily life in fibromyalgia patients.Scand J Pain. 2016 Oct;13:166-172. doi: 10.1016/j.sjpain.2016.07.004. Epub 2016 Aug 22. Scand J Pain. 2016. PMID: 28850525
-
Low fermentable oligo-di-mono-saccharides and polyols diet versus general dietary advice in patients with diarrhea-predominant irritable bowel syndrome: A randomized controlled trial.J Gastroenterol Hepatol. 2018 Jun;33(6):1192-1199. doi: 10.1111/jgh.14051. Epub 2018 Feb 21. J Gastroenterol Hepatol. 2018. PMID: 29159993 Clinical Trial.
-
Consumption of a Low Fermentable Oligo-, Di-, Mono-saccharides, and Polyols Diet and Irritable Bowel Syndrome: A Systematic Review.Int J Prev Med. 2017 Dec 13;8:104. doi: 10.4103/ijpvm.IJPVM_175_17. eCollection 2017. Int J Prev Med. 2017. PMID: 29416833 Free PMC article. Review.
-
Efficacy of a low-FODMAP diet in adult irritable bowel syndrome: a systematic review and meta-analysis.Eur J Nutr. 2021 Sep;60(6):3505-3522. doi: 10.1007/s00394-020-02473-0. Epub 2021 Feb 14. Eur J Nutr. 2021. PMID: 33585949 Free PMC article.
Cited by
-
Work-related problems and the psychosocial characteristics of individuals with irritable bowel syndrome: an updated literature review.Biopsychosoc Med. 2024 May 15;18(1):12. doi: 10.1186/s13030-024-00309-5. Biopsychosoc Med. 2024. PMID: 38750514 Free PMC article. Review.
-
Food Literacy and Its Associated Factors in Non-Dialysis Patients with Chronic Kidney Disease in China: A Cross-Sectional Study.Patient Prefer Adherence. 2022 Feb 18;16:439-447. doi: 10.2147/PPA.S348227. eCollection 2022. Patient Prefer Adherence. 2022. PMID: 35210760 Free PMC article.
-
Understanding the user: Patients' perception, needs, and concerns of health apps for chronic constipation.Digit Health. 2022 May 29;8:20552076221104673. doi: 10.1177/20552076221104673. eCollection 2022 Jan-Dec. Digit Health. 2022. PMID: 35663236 Free PMC article.
-
mHealth Intervention for Improving Pain, Quality of Life, and Functional Disability in Patients With Chronic Pain: Systematic Review.JMIR Mhealth Uhealth. 2023 Feb 2;11:e40844. doi: 10.2196/40844. JMIR Mhealth Uhealth. 2023. PMID: 36729570 Free PMC article.
-
Randomized Controlled Trials of Artificial Intelligence in Clinical Practice: Systematic Review.J Med Internet Res. 2022 Aug 25;24(8):e37188. doi: 10.2196/37188. J Med Internet Res. 2022. PMID: 35904087 Free PMC article.
References
-
- Sperber AD, Bangdiwala SI, Drossman DA, Ghoshal UC, Simren M, Tack J, Whitehead WE, Dumitrascu DL, Fang X, Fukudo S, Kellow J, Okeke E, Quigley EMM, Schmulson M, Whorwell P, Archampong T, Adibi P, Andresen V, Benninga MA, Bonaz B, Bor S, Fernandez LB, Choi SC, Corazziari ES, Francisconi C, Hani A, Lazebnik L, Lee YY, Mulak A, Rahman MM, Santos J, Setshedi M, Syam AF, Vanner S, Wong RK, Lopez-Colombo A, Costa V, Dickman R, Kanazawa M, Keshteli AH, Khatun R, Maleki I, Poitras P, Pratap N, Stefanyuk O, Thomson S, Zeevenhooven J, Palsson OS. Worldwide prevalence and burden of functional gastrointestinal disorders, results of Rome Foundation Global Study. Gastroenterology. 2021 Jan;160(1):99–114.e3. doi: 10.1053/j.gastro.2020.04.014. https://linkinghub.elsevier.com/retrieve/pii/S0016-5085(20)30487-X - DOI - PubMed
-
- Nyrop KA, Palsson OS, Levy RL, Von Korff M, Feld AD, Turner MJ, Whitehead WE. Costs of health care for irritable bowel syndrome, chronic constipation, functional diarrhoea and functional abdominal pain. Aliment Pharmacol Ther. 2007 Jul 15;26(2):237–48. doi: 10.1111/j.1365-2036.2007.03370.x. doi: 10.1111/j.1365-2036.2007.03370.x. - DOI - DOI - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

