Quantification of Hepatitis B Virus Covalently Closed Circular DNA in Infected Cell Culture Models by Quantitative PCR
- PMID: 33654998
- PMCID: PMC7854259
- DOI: 10.21769/BioProtoc.3202
Quantification of Hepatitis B Virus Covalently Closed Circular DNA in Infected Cell Culture Models by Quantitative PCR
Abstract
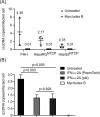
Persistence of the human hepatitis B virus (HBV) requires the maintenance of covalently closed circular (ccc)DNA, the episomal genome reservoir in nuclei of infected hepatocytes. cccDNA elimination is a major aim in future curative therapies currently under development. In cell culture based in vitro studies, both hybridization- and amplification-based assays are currently used for cccDNA quantification. Southern blot, the current gold standard, is time-consuming and not practical for a large number of samples. PCR-based methods show limited specificity when excessive HBV replicative intermediates are present. We have recently developed a real-time quantitative PCR protocol, in which total cellular DNA plus all forms of viral DNA are extracted by silica column. Subsequent incubation with T5 exonuclease efficiently removes cellular DNA and all non-cccDNA forms of viral DNA while cccDNA remains intact and can reliably be quantified by PCR. This method has been used for measuring kinetics of cccDNA accumulation in several in vitro infection models and the effect of antivirals on cccDNA. It allowed detection of cccDNA in non-human cells (primary macaque and swine hepatocytes, etc.) reconstituted with the HBV receptor, human sodium taurocholate cotransporting polypeptide (NTCP). Here we present a detailed protocol of this method, including a work flowchart, schematic diagram and illustrations on how to calculate "cccDNA copies per (infected) cell".
Keywords: Copy number per cell; Covalently closed circular DNA; Hepatitis B Virus; Quantitative PCR; T5 exonuclease; cccDNA.
Copyright © 2019 The Authors; exclusive licensee Bio-protocol LLC.
Conflict of interest statement
Competing interestsProf. Dr. Stephan Urban, the corresponding author, holds patents and intellectual property on Myrcludex B. Ethics Following written informed consent of the patients, PHH were isolated from liver specimens obtained after partial hepatectomy.
Figures


References
-
- Lempp F. A., Wiedtke E., Qu B., Roques P., Chemin I., Vondran F. W. R., Le Grand R., Grimm D. and Urban S.(2017). Sodium taurocholate cotransporting polypeptide is the limiting host factor of hepatitis B virus infection in macaque and pig hepatocytes. Hepatology 66(3): 703-716. - PubMed
LinkOut - more resources
Full Text Sources

