Extracellular Pneumococcal Serine Proteases Affect Nasopharyngeal Colonization
- PMID: 33659218
- PMCID: PMC7917122
- DOI: 10.3389/fcimb.2020.613467
Extracellular Pneumococcal Serine Proteases Affect Nasopharyngeal Colonization
Abstract
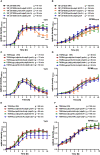
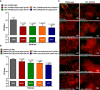
Streptococcus pneumoniae has evolved versatile strategies to colonize the nasopharynx of humans. Colonization is facilitated by direct interactions with host cell receptors or via binding to components of the extracellular matrix. In addition, pneumococci hijack host-derived extracellular proteases such as the serine protease plasmin(ogen) for ECM and mucus degradation as well as colonization. S. pneumoniae expresses strain-dependent up to four serine proteases. In this study, we assessed the role of secreted or cell-bound serine proteases HtrA, PrtA, SFP, and CbpG, in adherence assays and in a mouse colonization model. We hypothesized that the redundancy of serine proteases compensates for the deficiency of a single enzyme. Therefore, double and triple mutants were generated in serotype 19F strain EF3030 and serotype 4 strain TIGR4. Strain EF3030 produces only three serine proteases and lacks the SFP encoding gene. In adherence studies using Detroit-562 epithelial cells, we demonstrated that both TIGR4Δcps and 19F mutants without serine proteases or expressing only CbpG, HtrA, or PrtA have a reduced ability to adhere to Detroit-562 cells. Consistent with these results, we show that the mutants of strain 19F, which preferentially colonizes mice, abrogate nasopharyngeal colonization in CD-1 mice after intranasal infection. The bacterial load in the nasopharynx was monitored for 14 days. Importantly, mutants showed significantly lower bacterial numbers in the nasopharynx two days after infection. Similarly, we detected a significantly reduced pneumococcal colonization on days 3, 7, and 14 post-inoculations. To assess the impact of pneumococcal serine proteases on acute infection, we infected mice intranasally with bioluminescent and invasive TIGR4 or isogenic triple mutants expressing only CbpG, HtrA, PrtA, or SFP. We imaged the acute lung infection in real-time and determined the survival of the mice. The TIGR4lux mutant expressing only PrtA showed a significant attenuation and was less virulent in the acute pneumonia model. In conclusion, our results showed that pneumococcal serine proteases contributed significantly to pneumococcal colonization but played only a minor role in pneumonia and invasive diseases. Because colonization is a prerequisite for invasive diseases and transmission, these enzymes could be promising candidates for the development of antimicrobials to reduce pneumococcal transmission.
Keywords: adherence; colonization; pneumococci; pneumonia; serine proteases.
Copyright © 2021 Ali, Kohler, Burchhardt, Wüst, Henck, Bolsmann, Voß and Hammerschmidt.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



Similar articles
-
Pneumococcal Extracellular Serine Proteases: Molecular Analysis and Impact on Colonization and Disease.Front Cell Infect Microbiol. 2021 Nov 1;11:763152. doi: 10.3389/fcimb.2021.763152. eCollection 2021. Front Cell Infect Microbiol. 2021. PMID: 34790590 Free PMC article. Review.
-
Streptococcus pneumoniae serine protease HtrA, but not SFP or PrtA, is a major virulence factor in pneumonia.PLoS One. 2013 Nov 11;8(11):e80062. doi: 10.1371/journal.pone.0080062. eCollection 2013. PLoS One. 2013. PMID: 24244609 Free PMC article.
-
Multifunctional role of choline binding protein G in pneumococcal pathogenesis.Infect Immun. 2006 Feb;74(2):821-9. doi: 10.1128/IAI.74.2.821-829.2006. Infect Immun. 2006. PMID: 16428724 Free PMC article.
-
Contributions of pneumolysin, pneumococcal surface protein A (PspA), and PspC to pathogenicity of Streptococcus pneumoniae D39 in a mouse model.Infect Immun. 2007 Apr;75(4):1843-51. doi: 10.1128/IAI.01384-06. Epub 2007 Jan 29. Infect Immun. 2007. PMID: 17261599 Free PMC article.
-
Pneumococci in biofilms are non-invasive: implications on nasopharyngeal colonization.Front Cell Infect Microbiol. 2014 Nov 6;4:163. doi: 10.3389/fcimb.2014.00163. eCollection 2014. Front Cell Infect Microbiol. 2014. PMID: 25414838 Free PMC article. Review.
Cited by
-
Intranasal trivalent candidate vaccine elicits broad humoral and cellular immunity against pneumococcal pneumonia.Front Cell Infect Microbiol. 2025 Jun 27;15:1563661. doi: 10.3389/fcimb.2025.1563661. eCollection 2025. Front Cell Infect Microbiol. 2025. PMID: 40654579 Free PMC article.
-
The global proteome of Streptococcus pneumoniae EF3030 under nutrient-defined in vitro conditions.Front Cell Infect Microbiol. 2025 Jul 11;15:1606161. doi: 10.3389/fcimb.2025.1606161. eCollection 2025. Front Cell Infect Microbiol. 2025. PMID: 40718681 Free PMC article.
-
Pneumococcal Extracellular Serine Proteases: Molecular Analysis and Impact on Colonization and Disease.Front Cell Infect Microbiol. 2021 Nov 1;11:763152. doi: 10.3389/fcimb.2021.763152. eCollection 2021. Front Cell Infect Microbiol. 2021. PMID: 34790590 Free PMC article. Review.
-
PneumoBrowse 2: an integrated visual platform for curated genome annotation and multiomics data analysis of Streptococcus pneumoniae.Nucleic Acids Res. 2025 Jan 6;53(D1):D839-D851. doi: 10.1093/nar/gkae923. Nucleic Acids Res. 2025. PMID: 39436044 Free PMC article.
-
New Insights into Beta-Lactam Resistance of Streptococcus pneumoniae: Serine Protease HtrA Degrades Altered Penicillin-Binding Protein 2x.Microorganisms. 2021 Aug 8;9(8):1685. doi: 10.3390/microorganisms9081685. Microorganisms. 2021. PMID: 34442764 Free PMC article.
References
-
- Andersson B., Dahmén J., Frejd T., Leffler H., Magnusson G., Noori G., et al. . (1983). Identification of an active disaccharide unit of a glycoconjugate receptor for pneumococci attaching to human pharyngeal epithelial cells. J. Exp. Med. 158 (2), 559–570. 10.1084/jem.158.2.559 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

