Optimization of dose and route of administration of the P-glycoprotein inhibitor, valspodar (PSC-833) and the P-glycoprotein and breast cancer resistance protein dual-inhibitor, elacridar (GF120918) as dual infusion in rats
- PMID: 33660938
- PMCID: PMC7931226
- DOI: 10.1002/prp2.740
Optimization of dose and route of administration of the P-glycoprotein inhibitor, valspodar (PSC-833) and the P-glycoprotein and breast cancer resistance protein dual-inhibitor, elacridar (GF120918) as dual infusion in rats
Abstract
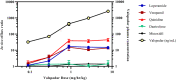
Transporters can play a key role in the absorption, distribution, metabolism, and excretion of drugs. Understanding these contributions early in drug discovery allows for more accurate projection of the clinical pharmacokinetics. One method to assess the impact of transporters in vivo involves co-dosing specific inhibitors. The objective of the present study was to optimize the dose and route of administration of a P-glycoprotein (P-gp) inhibitor, valspodar (PSC833), and a dual P-gp/breast cancer resistance protein (BCRP) inhibitor, elacridar (GF120918), by assessing the transporters' impact on brain penetration and absorption. A dual-infusion strategy was implemented to allow for flexibility with dose formulation. The chemical inhibitor was dosed intravenously via the femoral artery, and a cassette of known substrates was infused via the jugular vein. Valspodar or elacridar was administered as 4.5-hour constant infusions over a range of doses. To assess the degree of inhibition, the resulting ratios of brain and plasma concentrations, Kp's, of the known substrates were compared to the vehicle control. These data demonstrated that doses greater than 0.9 mg/hr/kg valspodar and 8.9 mg/hr/kg elacridar were sufficient to inhibit P-gp- and BCRP-mediated efflux at the blood-brain barrier in rats without any tolerability issues. Confirmation of BBB restriction by efflux transporters in preclinical species allows for subsequent prediction in humans based upon the proteomic expression at rodent and human BBB. Overall, the approach can also be applied to inhibition of efflux at other tissues (gut absorption, liver clearance) or can be extended to other transporters of interest using alternate inhibitors.
Keywords: BCRP; Breast cancer resistance protein; IVIVC; Kp; P-glycoprotein; P-gp; blood-brain barrier; chemical and genetic knock out; dantrolene; efflux transporter; elacridar; glyburide; loperamide; quinidine; rat; valspodar; verapamil.
© 2021 The Authors. Pharmacology Research & Perspectives published by John Wiley & Sons Ltd, British Pharmacological Society and American Society for Pharmacology and Experimental Therapeutics.
Conflict of interest statement
The authors have no conflicts of interest to declare. All co‐authors have seen and agree with the contents of the manuscript and there is no financial interest to report. We certify that the submission is original work and is not under review at any other publication.
Figures





References
-
- Uchida Y, Ohtsuki S, Kamiie J, Terasaki T. Blood‐brain barrier (BBB) pharmacoproteomics: reconstruction of in vivo brain distribution of 11 P‐glycoprotein substrates based on the BBB transporter protein concentration, in vitro intrinsic transport activity, and unbound fraction in plasma and brain in mice. J Pharmacol Exp Ther. 2011;339(2):579‐588. - PubMed
-
- Chu X, Zhang Z, Yabut J, et al. Characterization of multidrug resistance 1a/P‐glycoprotein knockout rats generated by zinc finger nucleases. Mol Pharmacol. 2012;81(2):220‐227. - PubMed
-
- Dufek MB, Knight BM, Bridges AS, Thakker DR. P‐glycoprotein increases portal bioavailability of loperamide in mouse by reducing first‐pass intestinal metabolism. Drug Metab Dispos. 2013;41(3):642‐650. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

