Hepcidin-Mediated Hypoferremia Disrupts Immune Responses to Vaccination and Infection
- PMID: 33665641
- PMCID: PMC7895906
- DOI: 10.1016/j.medj.2020.10.004
Hepcidin-Mediated Hypoferremia Disrupts Immune Responses to Vaccination and Infection
Abstract
Background: How specific nutrients influence adaptive immunity is of broad interest. Iron deficiency is the most common micronutrient deficiency worldwide and imparts a significant burden of global disease; however, its effects on immunity remain unclear.
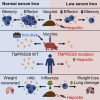
Methods: We used a hepcidin mimetic and several genetic models to examine the effect of low iron availability on T cells in vitro and on immune responses to vaccines and viral infection in mice. We examined humoral immunity in human patients with raised hepcidin and low serum iron caused by mutant TMPRSS6. We tested the effect of iron supplementation on vaccination-induced humoral immunity in piglets, a natural model of iron deficiency.
Findings: We show that low serum iron (hypoferremia), caused by increased hepcidin, severely impairs effector and memory responses to immunizations. The intensified metabolism of activated lymphocytes requires the support of enhanced iron acquisition, which is facilitated by IRP1/2 and TFRC. Accordingly, providing extra iron improved the response to vaccination in hypoferremic mice and piglets, while conversely, hypoferremic humans with chronically increased hepcidin have reduced concentrations of antibodies specific for certain pathogens. Imposing hypoferremia blunted the T cell, B cell, and neutralizing antibody responses to influenza virus infection in mice, allowing the virus to persist and exacerbating lung inflammation and morbidity.
Conclusions: Hypoferremia, a well-conserved physiological innate response to infection, can counteract the development of adaptive immunity. This nutrient trade-off is relevant for understanding and improving immune responses to infections and vaccines in the globally common contexts of iron deficiency and inflammatory disorders.
Funding: Medical Research Council, UK.
Keywords: T-cells; adaptive immunity; global health; hepcidin; hypoferremia; immunometabolism; infection; influenza virus; iron; vaccination.
© 2020 The Authors.
Conflict of interest statement
H.D. has sat on the advisory board of Kymab, received research funding from Pfizer and La Jolla Pharmaceutical Company, and received honoraria from Pharmacosmos and Vifor. The other authors declare no competing interests.
Figures







Comment in
-
Ironing Out Vaccine Efficacy.Med. 2021 Feb 12;2(2):113-114. doi: 10.1016/j.medj.2021.01.003. Med. 2021. PMID: 35187513 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

