Sodium-Glucose Co-Transporter 2 Inhibitors (SGLT2i) Exposure and Outcomes in Type 2 Diabetes: A Systematic Review of Population-Based Observational Studies
- PMID: 33665777
- PMCID: PMC7994468
- DOI: 10.1007/s13300-021-01004-2
Sodium-Glucose Co-Transporter 2 Inhibitors (SGLT2i) Exposure and Outcomes in Type 2 Diabetes: A Systematic Review of Population-Based Observational Studies
Abstract
Introduction: Sodium-glucose co-transporter 2 inhibitors (SGLT2is) are licensed for the treatment of type 2 diabetes (T2D) and more recently for heart failure with or without diabetes. They have been shown to be safe (from the cardiovascular (CV) perspective) and effective (in terms of glycaemia, and in some cases, in reducing CV events) in extensive randomised controlled trials (RCTs). However, there remain concerns regarding the generalisability of these findings (to those ineligible for RCT participation) and about non-CV safety. For effectiveness, population-based pharmacoepidemiology studies can confirm and extend the findings of RCTs to broader populations and explore safety, for which RCTs are not usually powered, in more detail.
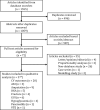
Methods: A pre-planned and registered ((International PROSPEctive Register Of Systematic Reviews) PROSPERO registration CRD42019160792) systematic review of population-based studies investigating SGLT2i effectiveness and safety, following Meta-analyses Of Observational Studies in Epidemiology (MOOSE) guidelines was conducted.
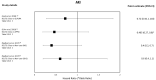
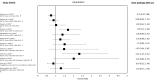
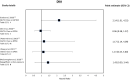
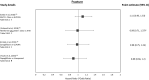
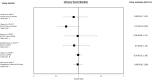
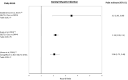
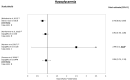
Results: A total of 37 studies were identified (total n = 1,300,184 adults; total follow-up 910,577 person-years; exposures: SGLT2i class, canagliflozin, dapagliflozin and empagliflozin) exploring CV disease (CVD) outcomes, acute kidney injury (AKI), lower limb amputation (LLA), diabetic ketoacidosis (DKA), bone fracture, urinary tract infection (UTI), genital mycotic infection (GMI), hypoglycaemia, pancreatitis and venous thromboembolism. For CV and mortality outcomes, studies confirmed the associated safety of these drugs and correlated closely with the findings from RCTs, which may extend to primary CVD prevention (major adverse cardiovascular events point estimate range (PER) hazard ratio (HR) 0.78-0.94; hospitalised heart failure PER HR 0.48-0.79). For safety outcomes, SGLT2i exposure was not associated with an increased risk of AKI (PER HR 0.40-0.96), fractures (PER HR 0.87-1.11), hypoglycaemia (PER HR 0.76-2.49) or UTI (PER HR 0.72-0.98). There was a signal for increased association for GMIs (PER HR 2.08-3.15), and possibly for LLA (PER HR 0.74-2.79) and DKA (PER HR 0.96-2.14), but with considerable uncertainty.
Conclusion: In T2D, SGLT2is appear safe from the CV perspective and may have associated benefit in primary as well as secondary CVD prevention. For safety, they may be associated with an increased risk of GMI, LLA and DKA, although longer follow-up studies are needed.
Keywords: Comparative effectiveness; Observational studies; SGLT2 inhibitors; Systematic review; Type 2 diabetes.
Figures
References
-
- Verma S, McMurray JJV. SGLT2 inhibitors and mechanisms of cardiovascular benefit: a state-of-the-art review. Diabetologia. 2018;61:2108–2117. - PubMed
-
- Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. NEJM. 2015;373:2117–2128. - PubMed
-
- Wiviott SD, Raz I, Bonaca MP, et al. Dapagliflozin and cardiovascular outcomes in type 2 diabetes. NEJM. 2019;380:347–357. - PubMed
-
- McMurray JJV, Solomon SD, Inzucchi SE, et al. Dapagliflozin in patients with heart failure and reduced ejection fraction. NEJM. 2019;381:1995–2008. - PubMed
-
- Neal B, Perkovic V, Mahaffey KW, et al. Canagliflozin and cardiovascular and renal events in type 2 diabetes. NEJM. 2017;377:644–657. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources