MKL1 cooperates with p38MAPK to promote vascular senescence, inflammation, and abdominal aortic aneurysm
- PMID: 33667992
- PMCID: PMC7937568
- DOI: 10.1016/j.redox.2021.101903
MKL1 cooperates with p38MAPK to promote vascular senescence, inflammation, and abdominal aortic aneurysm
Abstract
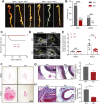
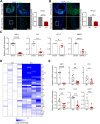
Abdominal aortic aneurysm (AAA) is a catastrophic disease with little effective therapy. Myocardin related transcription factor A (MRTFA, MKL1) is a multifaceted transcription factor, regulating diverse biological processes. However, a detailed understanding of the mechanistic role of MKL1 in AAA has yet to be elucidated. In this study, we showed induced MKL1 expression in thoracic and abdominal aneurysmal tissues, respectively in both mice and humans. MKL1 global knockout mice displayed reduced AAA formation and aortic rupture compared with wild-type mice. Both gene deletion and pharmacological inhibition of MKL1 markedly protected mice from aortic dissection, an early event in Angiotensin II (Ang II)-induced AAA formation. Loss of MKL1 was accompanied by reduced senescence/proinflammation in the vessel wall and cultured vascular smooth muscle cells (VSMCs). Mechanistically, a deficiency in MKL1 abolished AAA-induced p38 mitogen activated protein kinase (p38MAPK) activity. Similar to MKL1, loss of MAPK14 (p38α), the dominant isoform of p38MAPK family in VSMCs suppressed Ang II-induced AAA formation, vascular inflammation, and senescence marker expression. These results reveal a molecular pathway of AAA formation involving MKL1/p38MAPK stimulation and a VSMC senescent/proinflammatory phenotype. These data support targeting MKL1/p38MAPK pathway as a potential effective treatment for AAA.
Keywords: Aortic aneurysm; Inflammation; MKL1; Senescence; p38MAPK.
Copyright © 2021 The Authors. Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
None. Dr. LeMaire serves as a consultant for Terumo Aortic and Baxter Healthcare; serves as a principal investigator for clinical studies sponsored by Terumo Aortic and CytoSorbants; and serves as a co-investigator for clinical studies sponsored by W.L. Gore & Associates.
Figures







References
-
- Sakalihasan N., Limet R., Defawe O.D. Abdominal aortic aneurysm. Lancet. 2005;365:1577–1589. - PubMed
-
- Nordon I.M., Hinchliffe R.J., Loftus I.M., Thompson M.M. Pathophysiology and epidemiology of abdominal aortic aneurysms. Nat. Rev. Cardiol. 2011;8:92–102. - PubMed
-
- Abdominal aortic aneurysms. Nat. Rev. Dis. Prim. 2018;4:35. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

