Differential Regulation of Human Serotonin Receptor Type 3A by Chanoclavine and Ergonovine
- PMID: 33668306
- PMCID: PMC7956620
- DOI: 10.3390/molecules26051211
Differential Regulation of Human Serotonin Receptor Type 3A by Chanoclavine and Ergonovine
Abstract
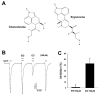
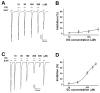
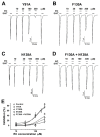
Irritable bowel syndrome (IBS) is a chronic disease that causes abdominal pain and an imbalance of defecation patterns due to gastrointestinal dysfunction. The cause of IBS remains unclear, but intestinal-brain axis problems and neurotransmitters have been suggested as factors. In this study, chanoclavine, which has a ring structure similar to 5-hydroxytryptamine (5-HT), showed an interaction with the 5-HT3A receptor to regulate IBS. Although its derivatives are known to be involved in neurotransmitter receptors, the molecular physiological mechanism of the interaction between chanoclavine and the 5-HT3A receptor is unknown. Electrophysiological experiments were conducted using a two-electrode voltage-clamp analysis to observe the inhibitory effects of chanoclavine on Xenopus oocytes in which the h5-HT3A receptor was expressed. The co-application of chanoclavine and 5-HT resulted in concentration-dependent, reversible, voltage-independent, and competitive inhibition. The 5-HT3A response induced by 5-HT was blocked by chanoclavine with half-maximal inhibitory response concentration (IC50) values of 107.2 µM. Docking studies suggested that chanoclavine was positioned close F130 and N138 in the 5-HT3A receptor-binding site. The double mutation of F130A and N138A significantly attenuated the interaction of chanoclavine compared to a single mutation or the wild type. These data suggest that F130 and N138 are important sites for ligand binding and activity. Chanoclavine and ergonovine have different effects. Asparagine, the 130th amino acid sequence of the 5-HT3A receptor, and phenylalanine, the 138th, are important in the role of binding chanoclavine, but ergonovine has no interaction with any amino acid sequence of the 5-HT3A receptor. The results of the electrophysiological studies and of in silico simulation showed that chanoclavine has the potential to inhibit the hypergastric stimulation of the gut by inhibiting the stimulation of signal transduction through 5-HT3A receptor stimulation. These findings suggest chanoclavine as a potential antiemetic agent for excessive gut stimulation and offer insight into the mechanisms of 5-HT3A receptor inhibition.
Keywords: chanoclavine; ergot alkaloids; irritable bowel syndrome; serotonin receptor.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Molecular Mechanisms of Nicergoline from Ergot Fungus in Blocking Human 5-HT3A Receptor.J Microbiol Biotechnol. 2024 Nov 29;35:e2411020. doi: 10.4014/jmb.2411.11020. J Microbiol Biotechnol. 2024. PMID: 39849925 Free PMC article.
-
Identification and molecular study on the interaction of Schisandrin C with human 5-HT3A receptor.Eur J Pharmacol. 2021 Sep 5;906:174220. doi: 10.1016/j.ejphar.2021.174220. Epub 2021 May 31. Eur J Pharmacol. 2021. PMID: 34081905
-
Differential effect of ginsenoside metabolites on the 5-HT3A receptor-mediated ion current in Xenopus oocytes.Mol Cells. 2004 Feb 29;17(1):51-6. Mol Cells. 2004. PMID: 15055527
-
Effects of ginsenoside Rg2 on the 5-HT3A receptor-mediated ion current in Xenopus oocytes.Mol Cells. 2003 Feb 28;15(1):108-13. Mol Cells. 2003. PMID: 12661769
-
Crosstalking interactions between P2X4 and 5-HT3A receptors.Neuropharmacology. 2023 Sep 15;236:109574. doi: 10.1016/j.neuropharm.2023.109574. Epub 2023 May 6. Neuropharmacology. 2023. PMID: 37156336 Review.
Cited by
-
Molecular Mechanisms of Nicergoline from Ergot Fungus in Blocking Human 5-HT3A Receptor.J Microbiol Biotechnol. 2024 Nov 29;35:e2411020. doi: 10.4014/jmb.2411.11020. J Microbiol Biotechnol. 2024. PMID: 39849925 Free PMC article.
-
Identification of 2,4-Di-tert-butylphenol as a Novel Agonist for Insect Odorant Receptors.Int J Mol Sci. 2023 Dec 22;25(1):220. doi: 10.3390/ijms25010220. Int J Mol Sci. 2023. PMID: 38203390 Free PMC article.
-
The Potential of Selected Plants and Their Biologically Active Molecules in the Treatment of Depression and Anxiety Disorders.Int J Mol Sci. 2025 Mar 6;26(5):2368. doi: 10.3390/ijms26052368. Int J Mol Sci. 2025. PMID: 40076986 Free PMC article. Review.
-
Enhancement of debitterness, water-solubility, and neuroprotective effects of naringin by transglucosylation.Appl Microbiol Biotechnol. 2023 Oct;107(20):6205-6217. doi: 10.1007/s00253-023-12709-8. Epub 2023 Aug 29. Appl Microbiol Biotechnol. 2023. PMID: 37642718
-
Neurotransmitter Dysfunction in Irritable Bowel Syndrome: Emerging Approaches for Management.J Clin Med. 2021 Jul 31;10(15):3429. doi: 10.3390/jcm10153429. J Clin Med. 2021. PMID: 34362210 Free PMC article. Review.
References
-
- Berde B. Ergot compounds: A synopsis. Adv. Biochem. Psychopharmacol. 1980;23:3–23. - PubMed
-
- Buchta M., Cvak L. Ergot alkaloids and other metabolites of the genus Claviceps. In: Křen V., Cvak L., editors. Ergot: The Genus Claviceps. Harwood Academic Publishers; Amsterdam, The Netherlands: 1999. pp. 173–200.
-
- Silberstein S.D. The pharmacology of ergotamine and dihydroergotamine. Headache. 1997;37:S15–S25. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

