A Robust Bioassay of the Human Bradykinin B2 Receptor that Extends Molecular and Cellular Studies: The Isolated Umbilical Vein
- PMID: 33668382
- PMCID: PMC7996148
- DOI: 10.3390/ph14030177
A Robust Bioassay of the Human Bradykinin B2 Receptor that Extends Molecular and Cellular Studies: The Isolated Umbilical Vein
Abstract
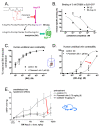
Bradykinin (BK) has various physiological and pathological roles. Medicinal chemistry efforts targeted toward the widely expressed BK B2 receptor (B2R), a G-protein-coupled receptor, were primarily aimed at developing antagonists. The only B2R antagonist in clinical use is the peptide icatibant, approved to abort attacks of hereditary angioedema. However, the anti-inflammatory applications of B2R antagonists are potentially wider. Furthermore, the B2R antagonists notoriously exhibit species-specific pharmacological profiles. Classical smooth muscle contractility assays are exploited over a time scale of several hours and support determining potency, competitiveness, residual agonist activity, specificity, and reversibility of pharmacological agents. The contractility assay based on the isolated human umbilical vein, expressing B2R at physiological density, was introduced when investigating the first non-peptide B2R antagonist (WIN 64338). Small ligand molecules characterized using the assay include the exquisitely potent competitive antagonist, Pharvaris Compound 3 or the partial agonist Fujisawa Compound 47a. The umbilical vein assay is also useful to verify pharmacologic properties of special peptide B2R ligands, such as the carboxypeptidase-activated latent agonists and fluorescent probes. Furthermore, the proposed agonist effect of tissue kallikrein on the B2R has been disproved using the vein. This assay stands in between cellular and molecular pharmacology and in vivo studies.
Keywords: B2 receptor; bradykinin; human umbilical vein; kallikrein-kinin system.
Conflict of interest statement
F.M. served as a consultant for Pharvaris B.V. and received research funds from Shire/Takeda and Pharvaris B.V.
Figures




Similar articles
-
D-Arg0-Bradykinin-Arg-Arg, a Latent Vasoactive Bradykinin B2 Receptor Agonist Metabolically Activated by Carboxypeptidases.Front Pharmacol. 2018 Mar 27;9:273. doi: 10.3389/fphar.2018.00273. eCollection 2018. Front Pharmacol. 2018. PMID: 29636689 Free PMC article.
-
Pharmacological effects of recombinant human tissue kallikrein on bradykinin B2 receptors.Pharmacol Res Perspect. 2015 Mar;3(2):e00119. doi: 10.1002/prp2.119. Epub 2015 Feb 10. Pharmacol Res Perspect. 2015. PMID: 26038695 Free PMC article.
-
Species-specific pharmacology of maximakinin, an amphibian homologue of bradykinin: putative prodrug activity at the human B2 receptor and peptidase resistance in rats.PeerJ. 2017 Jan 18;5:e2911. doi: 10.7717/peerj.2911. eCollection 2017. PeerJ. 2017. PMID: 28133580 Free PMC article.
-
Bifunctional ligands of the bradykinin B2 and B1 receptors: An exercise in peptide hormone plasticity.Peptides. 2018 Jul;105:37-50. doi: 10.1016/j.peptides.2018.05.007. Epub 2018 May 23. Peptides. 2018. PMID: 29802875 Review.
-
Targeting the 'Janus face' of the B2-bradykinin receptor.Expert Opin Ther Targets. 2013 Oct;17(10):1145-66. doi: 10.1517/14728222.2013.827664. Epub 2013 Aug 20. Expert Opin Ther Targets. 2013. PMID: 23957374 Review.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

