Cervid Prion Protein Polymorphisms: Role in Chronic Wasting Disease Pathogenesis
- PMID: 33668798
- PMCID: PMC7956812
- DOI: 10.3390/ijms22052271
Cervid Prion Protein Polymorphisms: Role in Chronic Wasting Disease Pathogenesis
Abstract
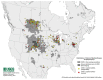
Chronic wasting disease (CWD) is a prion disease found in both free-ranging and farmed cervids. Susceptibility of these animals to CWD is governed by various exogenous and endogenous factors. Past studies have demonstrated that polymorphisms within the prion protein (PrP) sequence itself affect an animal's susceptibility to CWD. PrP polymorphisms can modulate CWD pathogenesis in two ways: the ability of the endogenous prion protein (PrPC) to convert into infectious prions (PrPSc) or it can give rise to novel prion strains. In vivo studies in susceptible cervids, complemented by studies in transgenic mice expressing the corresponding cervid PrP sequence, show that each polymorphism has distinct effects on both PrPC and PrPSc. It is not entirely clear how these polymorphisms are responsible for these effects, but in vitro studies suggest they play a role in modifying PrP epitopes crucial for PrPC to PrPSc conversion and determining PrPC stability. PrP polymorphisms are unique to one or two cervid species and most confer a certain degree of reduced susceptibility to CWD. However, to date, there are no reports of polymorphic cervid PrP alleles providing absolute resistance to CWD. Studies on polymorphisms have focused on those found in CWD-endemic areas, with the hope that understanding the role of an animal's genetics in CWD can help to predict, contain, or prevent transmission of CWD.
Keywords: cervid; chronic wasting disease; pathogenesis; polymorphism; prion protein; strain.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures


References
-
- Gilch S., Chitoor N., Taguchi Y., Stuart M., Jewell J.E., Schätzl H.M. Topics in Current Chemistry. Volume 305. Springer International Publishing; Cham, Switzerland: 2011. Chronic Wasting Disease; pp. 51–77. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

