Clade-Specific Plastid Inheritance Patterns Including Frequent Biparental Inheritance in Passiflora Interspecific Crosses
- PMID: 33668897
- PMCID: PMC7975985
- DOI: 10.3390/ijms22052278
Clade-Specific Plastid Inheritance Patterns Including Frequent Biparental Inheritance in Passiflora Interspecific Crosses
Abstract
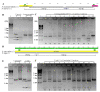
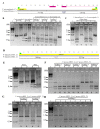
Plastid inheritance in angiosperms is presumed to be largely maternal, with the potential to inherit plastids biparentally estimated for about 20% of species. In Passiflora, maternal, paternal and biparental inheritance has been reported; however, these studies were limited in the number of crosses and progeny examined. To improve the understanding of plastid transmission in Passiflora, the progeny of 45 interspecific crosses were analyzed in the three subgenera: Passiflora, Decaloba and Astrophea. Plastid types were assessed following restriction digestion of PCR amplified plastid DNA in hybrid embryos, cotyledons and leaves at different developmental stages. Clade-specific patterns of inheritance were detected such that hybrid progeny from subgenera Passiflora and Astrophea predominantly inherited paternal plastids with occasional incidences of maternal inheritance, whereas subgenus Decaloba showed predominantly maternal and biparental inheritance. Biparental plastid inheritance was also detected in some hybrids from subgenus Passiflora. Heteroplasmy due to biparental inheritance was restricted to hybrid cotyledons and first leaves with a single parental plastid type detectable in mature plants. This indicates that in Passiflora, plastid retention at later stages of plant development may not reflect the plastid inheritance patterns in embryos. Passiflora exhibits diverse patterns of plastid inheritance, providing an excellent system to investigate underlying mechanisms in angiosperms.
Keywords: Passiflora; heteroplasmy; interspecific cross; plastid; plastid segregation/sorting-out; restriction digestion; uniparental and biparental inheritance.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Occurrence of plastids in the sperm cells of Caprifoliaceae: biparental plastid inheritance in angiosperms is unilaterally derived from maternal inheritance.Plant Cell Physiol. 2008 Jun;49(6):958-68. doi: 10.1093/pcp/pcn069. Epub 2008 Apr 29. Plant Cell Physiol. 2008. PMID: 18448473
-
Highly accelerated rates of genomic rearrangements and nucleotide substitutions in plastid genomes of Passiflora subgenus Decaloba.Mol Phylogenet Evol. 2019 Sep;138:53-64. doi: 10.1016/j.ympev.2019.05.030. Epub 2019 May 23. Mol Phylogenet Evol. 2019. PMID: 31129347
-
The model plant Medicago truncatula exhibits biparental plastid inheritance.Plant Cell Physiol. 2008 Jan;49(1):81-91. doi: 10.1093/pcp/pcm170. Epub 2007 Dec 6. Plant Cell Physiol. 2008. PMID: 18065422
-
Why does biparental plastid inheritance revive in angiosperms?J Plant Res. 2010 Mar;123(2):201-6. doi: 10.1007/s10265-009-0291-z. Epub 2010 Jan 7. J Plant Res. 2010. PMID: 20052516 Review.
-
Plastid Inheritance Revisited: Emerging Role of Organelle DNA Degradation in Angiosperms.Plant Cell Physiol. 2024 May 14;65(4):484-492. doi: 10.1093/pcp/pcad104. Plant Cell Physiol. 2024. PMID: 37702423 Review.
Cited by
-
Paternal leakage of plastids rescues inter-lineage hybrids in Silene nutans.Ann Bot. 2024 Apr 10;133(3):427-434. doi: 10.1093/aob/mcad196. Ann Bot. 2024. PMID: 38141228 Free PMC article.
-
The chloroplast genome inheritance pattern of the Deli-Nigerian prospection material (NPM) × Yangambi population of Elaeis guineensis Jacq.PeerJ. 2024 May 27;12:e17335. doi: 10.7717/peerj.17335. eCollection 2024. PeerJ. 2024. PMID: 38818457 Free PMC article.
-
Phylogenomic Inference Suggests Differential Deep Time Phylogenetic Signals from Nuclear and Organellar Genomes in Gymnosperms.Plants (Basel). 2025 Apr 28;14(9):1335. doi: 10.3390/plants14091335. Plants (Basel). 2025. PMID: 40364364 Free PMC article.
-
Cytoplasmic inheritance: The transmission of plastid and mitochondrial genomes across cells and generations.Plant Physiol. 2025 Apr 30;198(1):kiaf168. doi: 10.1093/plphys/kiaf168. Plant Physiol. 2025. PMID: 40304456 Free PMC article. Review.
-
Revealing the Introduction History and Phylogenetic Relationships of Passiflora foetida sensu lato in Australia.Front Plant Sci. 2021 Jul 29;12:651805. doi: 10.3389/fpls.2021.651805. eCollection 2021. Front Plant Sci. 2021. PMID: 34394135 Free PMC article.
References
-
- Kirk J.T.O., Tilney-Bassett R.A.E. The Plastids, Their Chemistry, Structure, Growth, and Inheritance. Elsevier/North Holland Biomedical Press; Amsterdam, The Netherlands: 1978.
-
- Birky C.W. Relaxed and stringent genomes: Why cytoplasmic genes don’t obey Mendel’s laws. J. Hered. 1994;85:355–365. doi: 10.1093/oxfordjournals.jhered.a111480. - DOI
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

