Monoassociation of Preterm Germ-Free Piglets with Bifidobacterium animalis Subsp. lactis BB-12 and Its Impact on Infection with Salmonella Typhimurium
- PMID: 33670419
- PMCID: PMC7917597
- DOI: 10.3390/biomedicines9020183
Monoassociation of Preterm Germ-Free Piglets with Bifidobacterium animalis Subsp. lactis BB-12 and Its Impact on Infection with Salmonella Typhimurium
Abstract
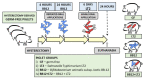
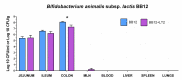
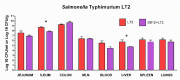
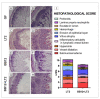
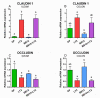
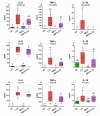
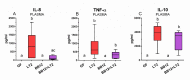
Preterm germ-free piglets were monoassociated with probiotic Bifidobacterium animalis subsp. lactis BB-12 (BB12) to verify its safety and to investigate possible protection against subsequent infection with Salmonella Typhimurium strain LT2 (LT2). Clinical signs of salmonellosis, bacterial colonization in the intestine, bacterial translocation to mesenteric lymph nodes (MLN), blood, liver, spleen, and lungs, histopathological changes in the ileum, claudin-1 and occludin mRNA expression in the ileum and colon, intestinal and plasma concentrations of IL-8, TNF-α, and IL-10 were evaluated. Both BB12 and LT2 colonized the intestine of the monoassociated piglets. BB12 did not translocate in the BB12-monoassociated piglets. BB12 was detected in some cases in the MLN of piglets, consequently infected with LT2, but reduced LT2 counts in the ileum and liver of these piglets. LT2 damaged the luminal structure of the ileum, but a previous association with BB12 mildly alleviated these changes. LT2 infection upregulated claudin-1 mRNA in the ileum and colon and downregulated occludin mRNA in the colon. Infection with LT2 increased levels of IL-8, TNF-α, and IL-10 in the intestine and plasma, and BB12 mildly downregulated them compared to LT2 alone. Despite reductions in bacterial translocation and inflammatory cytokines, clinical signs of LT2 infection were not significantly affected by the probiotic BB12. Thus, we hypothesize that multistrain bacterial colonization of preterm gnotobiotic piglets may be needed to enhance the protective effect against the infection with S. Typhimurium LT2.
Keywords: Bifidobacterium animalis subsp. lactis BB-12; Salmonella Typhimurium; immunocompromised; inflammatory cytokines; intestinal barrier; preterm host.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Release of HMGB1 and Toll-like Receptors 2, 4, and 9 Signaling Are Modulated by Bifidobacterium animalis subsp. lactis BB-12 and Salmonella Typhimurium in a Gnotobiotic Piglet Model of Preterm Infants.Int J Mol Sci. 2023 Jan 24;24(3):2329. doi: 10.3390/ijms24032329. Int J Mol Sci. 2023. PMID: 36768650 Free PMC article.
-
Impact of the Lipopolysaccharide Chemotype of Salmonella Enterica Serovar Typhimurium on Virulence in Gnotobiotic Piglets.Toxins (Basel). 2019 Sep 13;11(9):534. doi: 10.3390/toxins11090534. Toxins (Basel). 2019. PMID: 31540295 Free PMC article.
-
Xylo-oligosaccharide alleviates Salmonella induced inflammation by stimulating Bifidobacterium animalis and inhibiting Salmonella colonization.FASEB J. 2021 Nov;35(11):e21977. doi: 10.1096/fj.202100919RR. FASEB J. 2021. PMID: 34613640
-
Colonization of preterm gnotobiotic piglets with probiotic Lactobacillus rhamnosus GG and its interference with Salmonella Typhimurium.Clin Exp Immunol. 2019 Mar;195(3):381-394. doi: 10.1111/cei.13236. Epub 2018 Dec 2. Clin Exp Immunol. 2019. PMID: 30422309 Free PMC article.
-
The Science behind the Probiotic Strain Bifidobacterium animalis subsp. lactis BB-12(®).Microorganisms. 2014 Mar 28;2(2):92-110. doi: 10.3390/microorganisms2020092. Microorganisms. 2014. PMID: 27682233 Free PMC article. Review.
Cited by
-
Gut microbiota alteration with growth performance, histopathological lesions, and immune responses in Salmonella Typhimurium-infected weaned piglets.Vet Anim Sci. 2023 Nov 29;22:100324. doi: 10.1016/j.vas.2023.100324. eCollection 2023 Dec. Vet Anim Sci. 2023. PMID: 38125715 Free PMC article.
-
Release of HMGB1 and Toll-like Receptors 2, 4, and 9 Signaling Are Modulated by Bifidobacterium animalis subsp. lactis BB-12 and Salmonella Typhimurium in a Gnotobiotic Piglet Model of Preterm Infants.Int J Mol Sci. 2023 Jan 24;24(3):2329. doi: 10.3390/ijms24032329. Int J Mol Sci. 2023. PMID: 36768650 Free PMC article.
-
Defined Pig Microbiota with a Potential Protective Effect against Infection with Salmonella Typhimurium.Microorganisms. 2023 Apr 12;11(4):1007. doi: 10.3390/microorganisms11041007. Microorganisms. 2023. PMID: 37110429 Free PMC article.
-
Understanding the probiotic health benefits of Bifidobacterium animalis subsp. lactis, BB-12™.Front Microbiol. 2025 Jul 2;16:1605044. doi: 10.3389/fmicb.2025.1605044. eCollection 2025. Front Microbiol. 2025. PMID: 40673154 Free PMC article. Review.
-
Bifidobacterium animalis Promotes the Growth of Weaning Piglets by Improving Intestinal Development, Enhancing Antioxidant Capacity, and Modulating Gut Microbiota.Appl Environ Microbiol. 2022 Nov 22;88(22):e0129622. doi: 10.1128/aem.01296-22. Epub 2022 Oct 27. Appl Environ Microbiol. 2022. PMID: 36300953 Free PMC article.
References
-
- Milani C., Duranti S., Bottacini F., Casey E., Turroni F., Mahony J., Belzer C., Delgado P.S., Arboleya M.S., Mancabelli L., et al. The first microbial colonizers of the human gut: Composition, activities, and health implications of the infant gut microbiota. Microbiol. Mol. Biol. Rev. 2017;81 doi: 10.1128/MMBR.00036-17. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

