Development of a Specific Monoclonal Antibody to Detect Male Cells Expressing the RPS4Y1 Protein
- PMID: 33670450
- PMCID: PMC7921920
- DOI: 10.3390/ijms22042001
Development of a Specific Monoclonal Antibody to Detect Male Cells Expressing the RPS4Y1 Protein
Abstract
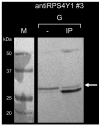
Hemophilia is an X-linked recessive bleeding disorder. In pregnant women carrier of hemophilia, the fetal sex can be determined by non-invasive analysis of fetal DNA circulating in the maternal blood. However, in case of a male fetus, conventional invasive procedures are required for the diagnosis of hemophilia. Fetal cells, circulating in the maternal bloodstream, are an ideal target for a safe non-invasive prenatal diagnosis. Nevertheless, the small number of cells and the lack of specific fetal markers have been the most limiting factors for their isolation. We aimed to develop monoclonal antibodies (mAbs) against the ribosomal protein RPS4Y1 expressed in male cells. By Western blotting, immunoprecipitation and immunofluorescence analyses performed on cell lysates from male human hepatoma (HepG2) and female human embryonic kidney (HEK293) we developed and characterized a specific monoclonal antibody against the native form of the male RPS4Y1 protein that can distinguish male from female cells. The availability of the RPS4Y1-targeting monoclonal antibody should facilitate the development of novel methods for the reliable isolation of male fetal cells from the maternal blood and their future use for non-invasive prenatal diagnosis of X-linked inherited disease such as hemophilia.
Keywords: RPS4Y1; diagnosis; fetal cells; hemophilia; male cells; male marker; monoclonal antibody; ribosomal protein.
Conflict of interest statement
F.P. has received honoraria for participating as a speaker at satellite symposia organized by Bioverativ, Grifols, Roche, Sanofi, Sobi, Spark and Takeda. F.P. reports participation at advisory board of Roche, Sanofi and Sobi. The authors declare no additional conflict of interest.
Figures




Similar articles
-
Identification of fetal gender in maternal blood is a helpful tool in the prenatal diagnosis of haemophilia.Haemophilia. 2006 Jul;12(4):417-22. doi: 10.1111/j.1365-2516.2006.01298.x. Haemophilia. 2006. PMID: 16834744
-
A comparison of the choice of monoclonal antibodies for recovery of fetal cells from maternal blood using FACS for noninvasive prenatal diagnosis of hemoglobinopathies.Cytometry B Clin Cytom. 2009 May;76(3):175-80. doi: 10.1002/cyto.b.20460. Cytometry B Clin Cytom. 2009. PMID: 18831520
-
Noninvasive prenatal diagnosis of hemophilia by microfluidics digital PCR analysis of maternal plasma DNA.Blood. 2011 Mar 31;117(13):3684-91. doi: 10.1182/blood-2010-10-310789. Epub 2011 Jan 24. Blood. 2011. PMID: 21263151
-
Recent advances in non-invasive prenatal DNA diagnosis through analysis of maternal blood.J Obstet Gynaecol Res. 2007 Dec;33(6):747-64. doi: 10.1111/j.1447-0756.2007.00652.x. J Obstet Gynaecol Res. 2007. PMID: 18001438 Review.
-
Prenatal screening of single-gene disorders from maternal blood.Am J Pharmacogenomics. 2001;1(2):111-7. doi: 10.2165/00129785-200101020-00004. Am J Pharmacogenomics. 2001. PMID: 12174672 Review.
Cited by
-
Ribosomal protein paralogues in ribosome specialization.Philos Trans R Soc Lond B Biol Sci. 2025 Mar 6;380(1921):20230387. doi: 10.1098/rstb.2023.0387. Epub 2025 Mar 6. Philos Trans R Soc Lond B Biol Sci. 2025. PMID: 40045786 Review.
-
Gender equity in hemophilia: need for healthcare, familial, and societal advocacy.Front Med (Lausanne). 2024 Apr 5;11:1345496. doi: 10.3389/fmed.2024.1345496. eCollection 2024. Front Med (Lausanne). 2024. PMID: 38646558 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

