New Insights into the Antimicrobial Action of Cinnamaldehyde towards Escherichia coli and Its Effects on Intestinal Colonization of Mice
- PMID: 33670478
- PMCID: PMC7922552
- DOI: 10.3390/biom11020302
New Insights into the Antimicrobial Action of Cinnamaldehyde towards Escherichia coli and Its Effects on Intestinal Colonization of Mice
Abstract
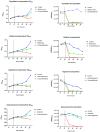
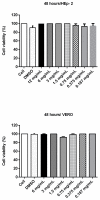
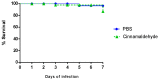
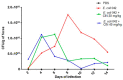
Escherichia coli is responsible for cases of diarrhea around the world, and some studies have shown the benefits of cinnamaldehyde in the treatment of bacterial disease. Therefore, the objective of this study was to evaluate the effects of cinnamaldehyde in mice colonized by pathogenic E. coli, as well as to provide more insights into its antimicrobial action mechanism. After determination of minimum inhibitory (MIC) and minimum bactericidal (MBC) concentrations, the interference of cinnamaldehyde in macromolecular pathways (synthesis of DNA, RNA, protein, and cell wall) was measured by incorporation of radioisotopes. The anti-adhesive properties of cinnamaldehyde towards E. coli 042 were evaluated using human epithelial type 2 (HEp-2) cells. Intestinal colonization was tested on mice, and the effect of cinnamaldehyde on Tenebrio molitor larvae. Cinnamaldehyde showed MIC and MBC values of 780 μg/mL and 1560 μg/mL, respectively; reduced the adhesion of E. coli 042 on HEp-2 cells; and affected all the synthetic pathways evaluated, suggesting that compost impairs the membrane/cell wall structure leading bacteria to total collapse. No effect on the expression of genes related to the SOS pathway (sulA and dinB1) was observed. The compound did not interfere with cell viability and was not toxic against T. molitor larvae. In addition, cinnamaldehyde-treated mice exhibited lower levels of colonization by E. coli 042 than the untreated group. Therefore, the results show that cinnamaldehyde is effective in treating the pathogenic E. coli strain 042 and confirm it as a promising lead molecule for the development of antimicrobial agents.
Keywords: cinnamaldehyde; intestinal colonization; natural products.
Conflict of interest statement
All authors: No reported conflicts.
Figures


References
-
- Taghadosi R., Shakibaie M.R., Hosseini-Nave H. Antibiotic resistance, ESBL genes, integrons, phylogenetic groups and MLVA profiles of Escherichia coli pathotypes isolated from patients with diarrhea and farm animals in south-east of Iran. Comp. Immunol. Microbiol. Infect. Dis. 2019;63:117–126. doi: 10.1016/j.cimid.2019.01.004. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

