Variables Affecting the Recovery of Acanthamoeba Trophozoites
- PMID: 33670669
- PMCID: PMC7922685
- DOI: 10.3390/pathogens10020221
Variables Affecting the Recovery of Acanthamoeba Trophozoites
Abstract
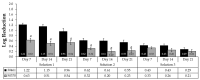
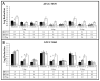
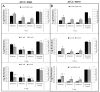
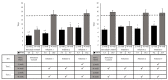
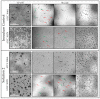
While the results of Acanthamoeba testing have been extensively published, laboratories conducting such testing are left to develop their own methods in the absence of a standardized methodology. The wide disparity of methods has resulted in equally inconsistent reported results for contact lens care (CLC) products. This study's objective was to determine the source of these discrepancies by evaluating basic Acanthamoeba biology and their impact on antimicrobial efficacy testing, including the ability of a recovery method to stimulate a single trophozoite to proliferate. Antimicrobial efficacy testing was conducted using well-published Acanthamoeba strains, storage conditions, and growth-based recovery methods. To identify variables that influence results, test solutions with low Acanthamoeba disinfection rates were utilized to prevent differences from being masked by high log reductions. In addition, single-cell proliferation assays were executed to understand the growth requirements to stimulate trophozoite propagation in two recovery methods. These studies indicated that both nutrient density (>106 CFU) and the length of plate incubation (at least 14 days) could significantly influence the accurate recovery of trophozoites. Together, this study emphasizes the need to understand how Acanthamoeba trophozoites biology can impact test methods to create divergent results.
Keywords: Acanthamoeba; contact lens care; efficacy testing; trophozoites.
Conflict of interest statement
All Authors are employees of Alcon Research, LLC.
Figures









Similar articles
-
Evaluating Alternate Methods of Determining the Antimicrobial Efficacy of Contact Lens Care Products against Acanthamoeba Trophozoites.Pathogens. 2021 Jan 27;10(2):126. doi: 10.3390/pathogens10020126. Pathogens. 2021. PMID: 33513702 Free PMC article.
-
Development of standardized methods for assessing biocidal efficacy of contact lens care solutions against Acanthamoeba trophozoites and cysts.Invest Ophthalmol Vis Sci. 2013 Jul 5;54(7):4527-37. doi: 10.1167/iovs.13-11927. Invest Ophthalmol Vis Sci. 2013. PMID: 23745008
-
Contact lens care solution killing efficacy against Acanthamoeba castellanii by in vitro testing and live-imaging.Cont Lens Anterior Eye. 2015 Dec;38(6):442-50. doi: 10.1016/j.clae.2015.06.006. Epub 2015 Jul 21. Cont Lens Anterior Eye. 2015. PMID: 26208952
-
Acanthamoeba: a review of its potential to cause keratitis, current lens care solution disinfection standards and methodologies, and strategies to reduce patient risk.Eye Contact Lens. 2008 Sep;34(5):247-53. doi: 10.1097/ICL.0b013e31817e7d83. Eye Contact Lens. 2008. PMID: 18779663 Review.
-
Acanthamoeba: An Overview of the Challenges to the Development of a Consensus Methodology of Disinfection Efficacy Testing for Contact Lens Care Products.Eye Contact Lens. 2018 Nov;44(6):351-354. doi: 10.1097/ICL.0000000000000470. Eye Contact Lens. 2018. PMID: 29697492 Review.
Cited by
-
Reduction of disinfection efficacy of contact lens care products on the global market in the presence of contact lenses and cases.BMJ Open Ophthalmol. 2022 Jun;7(1):e000955. doi: 10.1136/bmjophth-2021-000955. BMJ Open Ophthalmol. 2022. PMID: 36161836 Free PMC article.
-
Continuous Real-Time Motility Analysis of Acanthamoeba Reveals Sustained Movement in Absence of Nutrients.Pathogens. 2021 Aug 6;10(8):995. doi: 10.3390/pathogens10080995. Pathogens. 2021. PMID: 34451459 Free PMC article.
-
Differential Antimicrobial Efficacy of Preservative-Free Contact Lens Disinfection Systems against Common Ocular Pathogens.Microbiol Spectr. 2022 Feb 23;10(1):e0213821. doi: 10.1128/spectrum.02138-21. Epub 2022 Feb 9. Microbiol Spectr. 2022. PMID: 35138157 Free PMC article.
-
Complete Recovery of Acanthamoeba Motility among Surviving Organisms after Contact Lens Care Disinfection.Microorganisms. 2023 Jan 23;11(2):299. doi: 10.3390/microorganisms11020299. Microorganisms. 2023. PMID: 36838263 Free PMC article.
References
-
- Ahearn D.G., Gabriel M.M. Contact Lenses, Disinfectants, and Acanthamoeba Keratitis. Volume 43 Academic Press; Cambridge, MA, USA: 1997. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

