Comparison of the Efficacy of Two Novel Antitubercular Agents in Free and Liposome-Encapsulated Formulations
- PMID: 33671100
- PMCID: PMC7957691
- DOI: 10.3390/ijms22052457
Comparison of the Efficacy of Two Novel Antitubercular Agents in Free and Liposome-Encapsulated Formulations
Abstract
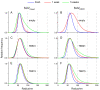
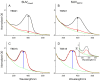
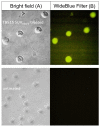
Tuberculosis is one of the top ten causes of death worldwide, and due to the appearance of drug-resistant strains, the development of new antituberculotic agents is a pressing challenge. Employing an in silico docking method, two coumaran (2,3-dihydrobenzofuran) derivatives-TB501 and TB515-were determined, with promising in vitro antimycobacterial activity. To enhance their effectiveness and reduce their cytotoxicity, we used liposomal drug carrier systems. Two types of small unilamellar vesicles (SUV) were prepared: multicomponent pH-sensitive stealth liposome (SUVmixed) and monocomponent conventional liposome. The long-term stability of our vesicles was obtained by the examination of particle size distribution with dynamic light scattering. Encapsulation efficiency (EE) of the two drugs was determined from absorption spectra before and after size exclusion chromatography. Cellular uptake and cytotoxicity were determined on human MonoMac-6 cells by flow cytometry. The antitubercular effect was characterized by the enumeration of colony-forming units on Mycobacterium tuberculosis H37Rv infected MonoMac-6 cultures. We found that SUVmixed + TB515 has the best long-term stability. TB515 has much higher EE in both types of SUVs. Cellular uptake for native TB501 is extremely low, but if it is encapsulated in SUVmixed it appreciably increases; in the case of TB515, quasi total uptake is accessible. It is concluded that SUVmixed + TB501 seems to be the most efficacious antitubercular formulation given the presented experiments; to find the most promising antituberculotic formulation for therapy further in vivo investigations are needed.
Keywords: 2,3-dihydrobenzofuran; absorption spectrometry; colony-forming units; coumaran; dynamic light scattering; flow cytometry; monocomponent liposome; pH-sensitive stealth liposome.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
[Development of antituberculous drugs: current status and future prospects].Kekkaku. 2006 Dec;81(12):753-74. Kekkaku. 2006. PMID: 17240921 Review. Japanese.
-
Antimycobacterial susceptibility evaluation of rifampicin and isoniazid benz-hydrazone in biodegradable polymeric nanoparticles against Mycobacterium tuberculosis H37Rv strain.Int J Nanomedicine. 2018 Jul 23;13:4303-4318. doi: 10.2147/IJN.S163925. eCollection 2018. Int J Nanomedicine. 2018. PMID: 30087562 Free PMC article.
-
Design of anti-bacterial drug and anti-mycobacterial drug for drug delivery system.Curr Pharm Des. 2002;8(6):475-82. doi: 10.2174/1381612023395808. Curr Pharm Des. 2002. PMID: 12069384
-
Enhanced cellular uptake of a new, in silico identified antitubercular candidate by peptide conjugation.Bioconjug Chem. 2012 May 16;23(5):900-7. doi: 10.1021/bc200221t. Epub 2012 Apr 19. Bioconjug Chem. 2012. PMID: 22515329
-
[Anti-tuberculosis drugs].Nihon Rinsho. 2007 Feb 28;65 Suppl 2 Pt. 1:367-74. Nihon Rinsho. 2007. PMID: 17455647 Review. Japanese. No abstract available.
Cited by
-
The modification of conventional liposomes for targeted antimicrobial delivery to treat infectious diseases.Discov Nano. 2025 Jan 30;20(1):19. doi: 10.1186/s11671-024-04170-x. Discov Nano. 2025. PMID: 39883380 Free PMC article. Review.
-
Optimization, characterization, and cytotoxicity studies of novel anti-tubercular agent-loaded liposomal vesicles.Sci Rep. 2024 Jan 4;14(1):524. doi: 10.1038/s41598-023-49576-2. Sci Rep. 2024. PMID: 38177202 Free PMC article.
-
Quantitative Analysis of a Pilot Transwell Barrier Model with Automated Sampling and Mathematical Modeling.Pharmaceutics. 2023 Nov 20;15(11):2646. doi: 10.3390/pharmaceutics15112646. Pharmaceutics. 2023. PMID: 38004624 Free PMC article.
-
Liposomal delivery system/adjuvant for tuberculosis vaccine.Immun Inflamm Dis. 2023 Jun;11(6):e867. doi: 10.1002/iid3.867. Immun Inflamm Dis. 2023. PMID: 37382263 Free PMC article. Review.
-
Liposome-Encapsulated Antibiotics for the Therapy of Mycobacterial Infections.Antibiotics (Basel). 2025 Jul 20;14(7):728. doi: 10.3390/antibiotics14070728. Antibiotics (Basel). 2025. PMID: 40724029 Free PMC article. Review.
References
-
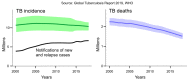
- World Health Organization . Global Tuberculosis Report 2019. World Health Organization; Geneva, Switzerland: 2019. Licence: CC BY-NC-SA 3.0 IGO.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

