Mast Cell and Astrocyte Hemichannels and Their Role in Alzheimer's Disease, ALS, and Harmful Stress Conditions
- PMID: 33672031
- PMCID: PMC7919494
- DOI: 10.3390/ijms22041924
Mast Cell and Astrocyte Hemichannels and Their Role in Alzheimer's Disease, ALS, and Harmful Stress Conditions
Abstract
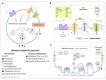
Considered relevant during allergy responses, numerous observations have also identified mast cells (MCs) as critical effectors during the progression and modulation of several neuroinflammatory conditions, including Alzheimer's disease (AD) and amyotrophic lateral sclerosis (ALS). MC granules contain a plethora of constituents, including growth factors, cytokines, chemokines, and mitogen factors. The release of these bioactive substances from MCs occurs through distinct pathways that are initiated by the activation of specific plasma membrane receptors/channels. Here, we focus on hemichannels (HCs) formed by connexins (Cxs) and pannexins (Panxs) proteins, and we described their contribution to MC degranulation in AD, ALS, and harmful stress conditions. Cx/Panx HCs are also expressed by astrocytes and are likely involved in the release of critical toxic amounts of soluble factors-such as glutamate, adenosine triphosphate (ATP), complement component 3 derivate C3a, tumor necrosis factor (TNFα), apoliprotein E (ApoE), and certain miRNAs-known to play a role in the pathogenesis of AD, ALS, and other neurodegenerative disorders. We propose that blocking HCs on MCs and glial cells offers a promising novel strategy for ameliorating the progression of neurodegenerative diseases by reducing the release of cytokines and other pro-inflammatory compounds.
Keywords: connexin; degranulation; gap junction channels; glial cells; hemichannels; inflammation; mast cells; neurodegeneration; pannexin; pro-inflammatory compounds.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Astroglial Connexins as a Therapeutic Target for Alzheimer's Disease.Curr Pharm Des. 2017;23(33):4958-4968. doi: 10.2174/1381612823666171004151215. Curr Pharm Des. 2017. PMID: 28982320 Review.
-
Regulation of pannexin and connexin channels and their functional role in skeletal muscles.Cell Mol Life Sci. 2015 Aug;72(15):2929-35. doi: 10.1007/s00018-015-1968-1. Epub 2015 Jun 18. Cell Mol Life Sci. 2015. PMID: 26084874 Free PMC article. Review.
-
Glial hemichannels and their involvement in aging and neurodegenerative diseases.Rev Neurosci. 2012 Jan 26;23(2):163-77. doi: 10.1515/revneuro-2011-0065. Rev Neurosci. 2012. PMID: 22499675 Review.
-
Pannexin-1 Channels Are Essential for Mast Cell Degranulation Triggered During Type I Hypersensitivity Reactions.Front Immunol. 2019 Nov 29;10:2703. doi: 10.3389/fimmu.2019.02703. eCollection 2019. Front Immunol. 2019. PMID: 31849935 Free PMC article.
-
Connexins and pannexins: At the junction of neuro-glial homeostasis & disease.J Neurosci Res. 2018 Jan;96(1):31-44. doi: 10.1002/jnr.24088. Epub 2017 Jun 5. J Neurosci Res. 2018. PMID: 28580666 Free PMC article. Review.
Cited by
-
Amyloid Beta Peptides Lead to Mast Cell Activation in a Novel 3D Hydrogel Model.Int J Mol Sci. 2023 Jul 26;24(15):12002. doi: 10.3390/ijms241512002. Int J Mol Sci. 2023. PMID: 37569378 Free PMC article.
-
Alternative Targets to Fight Alzheimer's Disease: Focus on Astrocytes.Biomolecules. 2021 Apr 19;11(4):600. doi: 10.3390/biom11040600. Biomolecules. 2021. PMID: 33921556 Free PMC article. Review.
-
Multifunctional regulation of VAMP3 in exocytic and endocytic pathways of RBL-2H3 cells.Front Immunol. 2022 Aug 5;13:885868. doi: 10.3389/fimmu.2022.885868. eCollection 2022. Front Immunol. 2022. PMID: 35990647 Free PMC article.
-
Tyrosine kinase inhibitor, masitinib, limits neuronal damage, as measured by serum neurofilament light chain concentration in a model of neuroimmune-driven neurodegenerative disease.PLoS One. 2025 May 14;20(5):e0322199. doi: 10.1371/journal.pone.0322199. eCollection 2025. PLoS One. 2025. PMID: 40367050 Free PMC article.
-
Impact of Mast Cell Activation on Neurodegeneration: A Potential Role for Gut-Brain Axis and Helicobacter pylori Infection.Neurol Int. 2024 Dec 6;16(6):1750-1778. doi: 10.3390/neurolint16060127. Neurol Int. 2024. PMID: 39728753 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

