Multiple Subtypes of Alzheimer's Disease Base on Brain Atrophy Pattern
- PMID: 33672406
- PMCID: PMC7926857
- DOI: 10.3390/brainsci11020278
Multiple Subtypes of Alzheimer's Disease Base on Brain Atrophy Pattern
Abstract
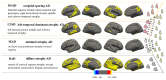
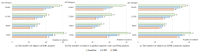
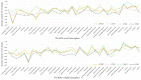
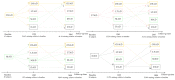
Alzheimer's disease (AD) is a disease of a heterogeneous nature, which can be disentangled by exploring the characteristics of each AD subtype in the brain structure, neuropathology, and cognition. In this study, a total of 192 AD and 228 cognitively normal (CN) subjects were obtained from the Alzheimer's disease Neuroimaging Initiative database. Based on the cortical thickness patterns, the mixture of experts method (MOE) was applied to the implicit model spectrum of transforms lined with each AD subtype, then their neuropsychological and neuropathological characteristics were analyzed. Furthermore, the piecewise linear classifiers composed of each AD subtype and CN were resolved, and each subtype was comprehensively explained. The following four distinct AD subtypes were discovered: bilateral parietal, frontal, and temporal atrophy AD subtype (occipital sparing AD subtype (OSAD), 29.2%), left temporal dominant atrophy AD subtype (LTAD, 22.4%), minimal atrophy AD subtype (MAD, 16.1%), and diffuse atrophy AD subtype (DAD, 32.3%). These four subtypes display their own characteristics in atrophy pattern, cognition, and neuropathology. Compared with the previous studies, our study found that some AD subjects showed obvious asymmetrical atrophy in left lateral temporal-parietal cortex, OSAD presented the worst cerebrospinal fluid levels, and MAD had the highest proportions of APOE ε4 and APOE ε2. The subtype characteristics were further revealed from the aspect of the model, making it easier for clinicians to understand. The results offer an effective support for individual diagnosis and prognosis.
Keywords: Alzheimer’s disease; atrophy subtypes; cortical thickness; mixture of experts; neuropathology; neuropsychology; structural magnetic resonance imaging.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Whitwell J.L., Dickson D.W., Murray M.E., Weigand S.D., Tosakulwong N., Senjem M.L., Knopman D.S., Boeve B.F., Parisi J.E., Petersen R.C., et al. Neuroimaging correlates of pathologically defined subtypes of Alzheimer’s disease: A case-control study. Lancet Neurol. 2012;11:868–877. doi: 10.1016/S1474-4422(12)70200-4. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

