Nrf2 Activation Sensitizes K-Ras Mutant Pancreatic Cancer Cells to Glutaminase Inhibition
- PMID: 33672789
- PMCID: PMC7918355
- DOI: 10.3390/ijms22041870
Nrf2 Activation Sensitizes K-Ras Mutant Pancreatic Cancer Cells to Glutaminase Inhibition
Abstract
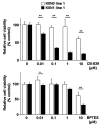
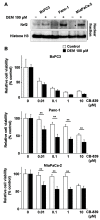
Pancreatic cancer remains intractable owing to the lack of effective therapy for unresectable cases. Activating mutations of K-ras are frequently found in pancreatic cancers, but these have not yet been targeted by cancer therapies. The Keap1-Nrf2 system plays a crucial role in mediating the oxidative stress response, which also contributes to cancer progression. Nrf2 activation reprograms the metabolic profile to promote the proliferation of cancer cells. A recent report suggested that K-ras- and Nrf2-active lung cancer cells are sensitive to glutamine depletion. This finding led to the recognition of glutaminase inhibitors as novel anticancer agents. In the current study, we used murine pancreatic cancer tissues driven by mutant K-ras and p53 to establish cell lines expressing constitutively activated Nrf2. Genetic or pharmacological Nrf2 activation in cells via Keap1 deletion or Nrf2 activation sensitized cells to glutaminase inhibition. This phenomenon was confirmed to be dependent on K-ras activation in human pancreatic cancer cell lines harboring mutant K-ras, i.e., Panc-1 and MiaPaCa-2 in response to DEM pretreatment. This phenomenon was not observed in BxPC3 cells harboring wildtype K-ras. These results indicate the possibility of employing Nrf2 activation and glutaminase inhibition as novel therapeutic interventions for K-ras mutant pancreatic cancers.
Keywords: BPTES; CB-839; Keap1; Nrf2; glutaminase.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Dematteo R.P., Ballman K.V., Antonescu C.R., Maki R.G., Pisters P.W., Demetri G.D., Blackstein M.E., Blanke C.D., von Mehren M., Brennan M.F., et al. Adjuvant imatinib mesylate after resection of localised, primary gastrointestinal stromal tumour: A randomised, double-blind, placebo-controlled trial. Lancet. 2009;373:1097–1104. doi: 10.1016/S0140-6736(09)60500-6. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

