Tumor Heterogeneity in Glioblastomas: From Light Microscopy to Molecular Pathology
- PMID: 33673104
- PMCID: PMC7918815
- DOI: 10.3390/cancers13040761
Tumor Heterogeneity in Glioblastomas: From Light Microscopy to Molecular Pathology
Abstract
One of the main reasons for the aggressive behavior of glioblastoma (GBM) is its intrinsic intra-tumor heterogeneity, characterized by the presence of clonal and subclonal differentiated tumor cell populations, glioma stem cells, and components of the tumor microenvironment, which affect multiple hallmark cellular functions in cancer. "Tumor Heterogeneity" usually encompasses both inter-tumor heterogeneity (population-level differences); and intra-tumor heterogeneity (differences within individual tumors). Tumor heterogeneity may be assessed in a single time point (spatial heterogeneity) or along the clinical evolution of GBM (longitudinal heterogeneity). Molecular methods may detect clonal and subclonal alterations to describe tumor evolution, even when samples from multiple areas are collected in the same time point (spatial-temporal heterogeneity). In GBM, although the inter-tumor mutational landscape is relatively homogeneous, intra-tumor heterogeneity is a striking feature of this tumor. In this review, we will address briefly the inter-tumor heterogeneity of the CNS tumors that yielded the current glioma classification. Next, we will take a deeper dive in the intra-tumor heterogeneity of GBMs, which directly affects prognosis and response to treatment. Our approach aims to follow technological developments, allowing for characterization of intra-tumor heterogeneity, beginning with differences on histomorphology of GBM and ending with molecular alterations observed at single-cell level.
Keywords: glioblastoma; glioma; neoplastic stem cells; precision medicine; prognosis; review; tumor heterogeneity; tumor microenvironment.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

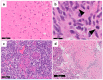



References
-
- Stupp R., Hegi M.E., Mason W.P., Bent M.J.V.D., Taphoorn M.J.B., Janzer R.C., Ludwin S.K., Allgeier A., Fisher B., Belanger K., et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 2009;10:459–466. doi: 10.1016/S1470-2045(09)70025-7. - DOI - PubMed
-
- Stupp R., Taillibert S., Kanner A., Read W., Steinberg D.M., Lhermitte B., Toms S., Idbaih A., Ahluwalia M.S., Fink K., et al. Effect of Tumor-Treating Fields Plus Maintenance Temozolomide vs Maintenance Temozolomide Alone on Survival in Patients With Glioblastoma. JAMA. 2017;318:2306–2316. doi: 10.1001/jama.2017.18718. - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

