Progress in the Development of Eukaryotic Elongation Factor 2 Kinase (eEF2K) Natural Product and Synthetic Small Molecule Inhibitors for Cancer Chemotherapy
- PMID: 33673713
- PMCID: PMC7957638
- DOI: 10.3390/ijms22052408
Progress in the Development of Eukaryotic Elongation Factor 2 Kinase (eEF2K) Natural Product and Synthetic Small Molecule Inhibitors for Cancer Chemotherapy
Abstract
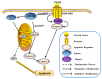
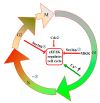
Eukaryotic elongation factor 2 kinase (eEF2K or Ca2+/calmodulin-dependent protein kinase, CAMKIII) is a new member of an atypical α-kinase family different from conventional protein kinases that is now considered as a potential target for the treatment of cancer. This protein regulates the phosphorylation of eukaryotic elongation factor 2 (eEF2) to restrain activity and inhibit the elongation stage of protein synthesis. Mounting evidence shows that eEF2K regulates the cell cycle, autophagy, apoptosis, angiogenesis, invasion, and metastasis in several types of cancers. The expression of eEF2K promotes survival of cancer cells, and the level of this protein is increased in many cancer cells to adapt them to the microenvironment conditions including hypoxia, nutrient depletion, and acidosis. The physiological function of eEF2K and its role in the development and progression of cancer are here reviewed in detail. In addition, a summary of progress for in vitro eEF2K inhibitors from anti-cancer drug discovery research in recent years, along with their structure-activity relationships (SARs) and synthetic routes or natural sources, is also described. Special attention is given to those inhibitors that have been already validated in vivo, with the overall aim to provide reference context for the further development of new first-in-class anti-cancer drugs that target eEF2K.
Keywords: cancer therapy; eEF2K; enzyme inhibitors; medicinal chemistry; natural products; protein kinase; small molecules.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this review.
Figures

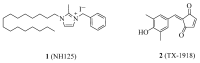

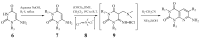





References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

