Bacteriophages against Vibrio coralliilyticus and Vibrio tubiashii: Isolation, Characterization, and Remediation of Larval Oyster Mortalities
- PMID: 33674441
- PMCID: PMC8117752
- DOI: 10.1128/AEM.00008-21
Bacteriophages against Vibrio coralliilyticus and Vibrio tubiashii: Isolation, Characterization, and Remediation of Larval Oyster Mortalities
Abstract
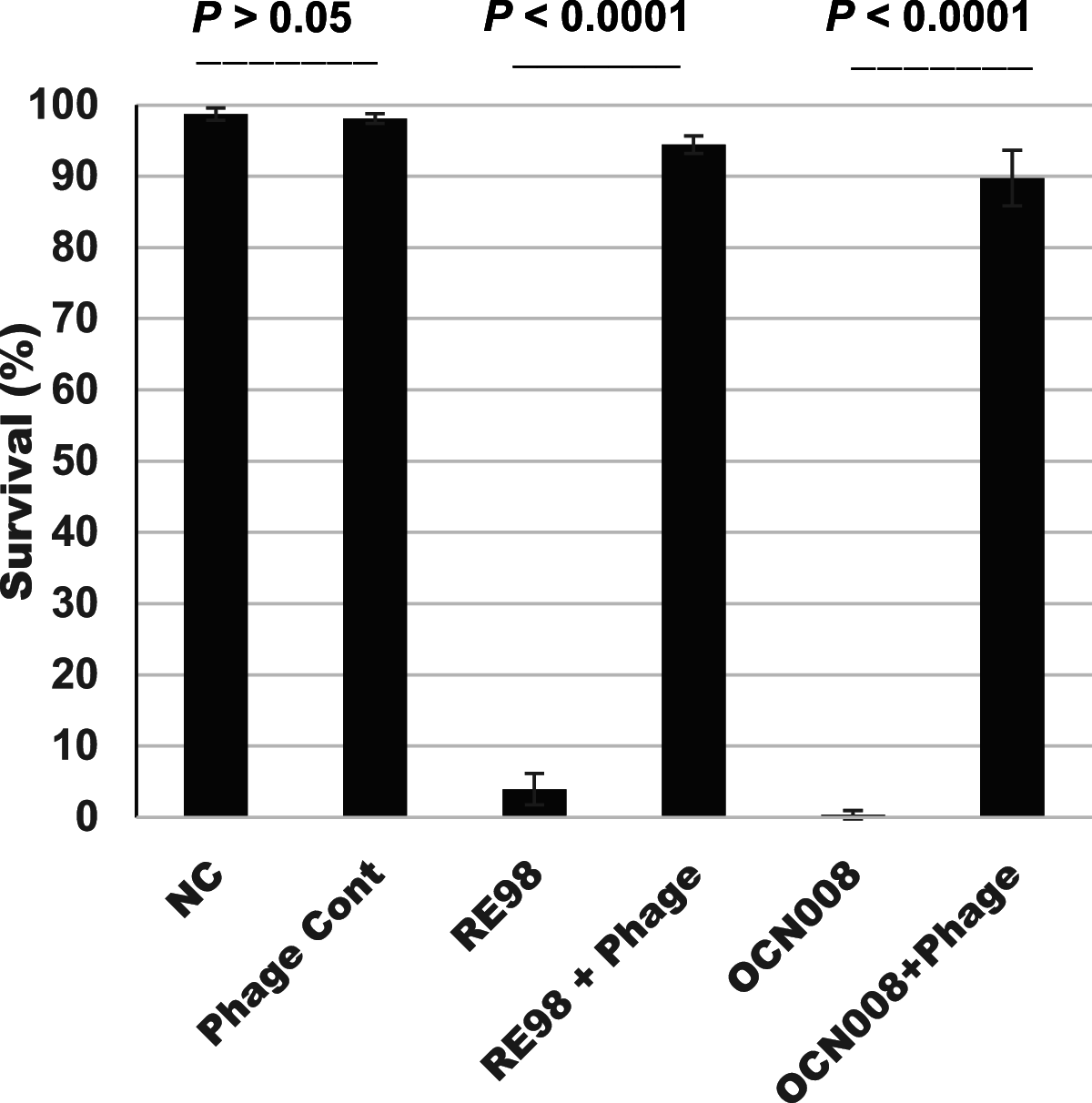
Vibrio coralliilyticus and Vibrio tubiashii are pathogens responsible for high larval oyster mortality rates in shellfish hatcheries. Bacteriophage therapy was evaluated to determine its potential to remediate these mortalities. Sixteen phages against V. coralliilyticus and V. tubiashii were isolated and characterized from Hawaiian seawater. Fourteen isolates were members of the Myoviridae family, and two were members of the Siphoviridae In proof-of-principle trials, a cocktail of five phages reduced mortalities of larval Eastern oysters (Crassostrea virginica) and Pacific oysters (Crassostrea gigas) by up to 91% 6 days after challenge with lethal doses of V. coralliilyticus Larval survival depended on the oyster species, the quantities of phages and vibrios applied, and the species and strain of Vibrio A later-generation cocktail, designated VCP300, was formulated with three lytic phages subsequently named Vibrio phages vB_VcorM-GR7B, vB_VcorM-GR11A, and vB_VcorM-GR28A (abbreviated 7B, 11A, and 28A, respectively). Together, these three phages displayed host specificity toward eight V. coralliilyticus strains and a V. tubiashii strain. Larval C. gigas mortalities from V. coralliilyticus strains RE98 and OCN008 were significantly reduced by >90% (P < 0.0001) over 6 days with phage treatment compared to those of untreated controls. Genomic sequencing of phages 7B, 11A, and 28A revealed 207,758-, 194,800-, and 154,046-bp linear DNA genomes, respectively, with the latter showing 92% similarity to V. coralliilyticus phage YC, a strain from the Great Barrier Reef, Australia. Phage 7B and 11A genomes showed little similarity to phages in the NCBI database. This study demonstrates the promising potential for phage therapy to reduce larval oyster mortalities in oyster hatcheries.IMPORTANCE Shellfish hatcheries encounter episodic outbreaks of larval oyster mortalities, jeopardizing the economic stability of hatcheries and the commercial shellfish industry. Shellfish pathogens like Vibrio coralliilyticus and Vibrio tubiashii have been recognized as major contributors of larval oyster mortalities in U.S. East and West Coast hatcheries for many years. This study isolated, identified, and characterized bacteriophages against these Vibrio species and demonstrated their ability to reduce mortalities from V. coralliilyticus in larval Pacific oysters and from both V. coralliilyticus and V. tubiashii in larval Eastern oysters. Phage therapy offers a promising approach for stimulating hatchery production to ensure the well-being of hatcheries and the commercial oyster trade.
Keywords: Vibrio coralliilyticus; Vibrio tubiashii; bacteriophage therapy; characterization; coral; genomic sequencing; isolation; larvae; mortalities; oyster; phage.
Copyright © 2021 American Society for Microbiology.
Figures



References
-
- Banks P, Geiger S, Herrmann J, Robertson C, Robinson L, VanderKooy S. 2014. Current regulations related to the propagation, transport, and release of hatchery raised oysters in the U.S. Gulf of Mexico. Gulf States Marine Fisheries Commission publication 238. Gulf States Marine Fisheries Commission, Ocean Springs, MS.
-
- Brown C. 1981. A study of two shellfish-pathogenic Vibrio strains isolated from a Long Island hatchery during a recent outbreak of disease. J Shellfish Res 1:83–87.
Publication types
MeSH terms
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

