Human type 1 and type 2 conventional dendritic cells express indoleamine 2,3-dioxygenase 1 with functional effects on T cell priming
- PMID: 33675038
- PMCID: PMC8251546
- DOI: 10.1002/eji.202048580
Human type 1 and type 2 conventional dendritic cells express indoleamine 2,3-dioxygenase 1 with functional effects on T cell priming
Abstract
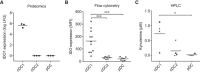
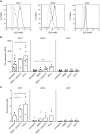
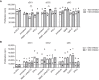
Dendritic cells (DCs) are key regulators of the immune system that shape T cell responses. Regulation of T cell induction by DCs may occur via the intracellular enzyme indoleamine 2,3-dioxygenase 1 (IDO), which catalyzes conversion of the essential amino acid tryptophan into kynurenine. Here, we examined the role of IDO in human peripheral blood plasmacytoid DCs (pDCs), and type 1 and type 2 conventional DCs (cDC1s and cDC2s). Our data demonstrate that under homeostatic conditions, IDO is selectively expressed by cDC1s. IFN-γ or TLR ligation further increases IDO expression in cDC1s and induces modest expression of the enzyme in cDC2s, but not pDCs. IDO expressed by conventional DCs is functionally active as measured by kynurenine production. Furthermore, IDO activity in TLR-stimulated cDC1s and cDC2s inhibits T cell proliferation in settings were DC-T cell cell-cell contact does not play a role. Selective inhibition of IDO1 with epacadostat, an inhibitor currently tested in clinical trials, rescued T cell proliferation without affecting DC maturation status or their ability to cross-present soluble antigen. Our findings provide new insights into the functional specialization of human blood DC subsets and suggest a possible synergistic enhancement of therapeutic efficacy by combining DC-based cancer vaccines with IDO inhibition.
Keywords: IDO; cDC1; cDC2; epacadostat; plasmacytoid dendritic cells.
© 2021 The Authors. European Journal of Immunology published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no commercial or financial conflict of interest.
Figures
Similar articles
-
The IDO1 selective inhibitor epacadostat enhances dendritic cell immunogenicity and lytic ability of tumor antigen-specific T cells.Oncotarget. 2016 Jun 21;7(25):37762-37772. doi: 10.18632/oncotarget.9326. Oncotarget. 2016. PMID: 27192116 Free PMC article.
-
Indoleamine 2,3-dioxygenase production by human dendritic cells results in the inhibition of T cell proliferation.J Immunol. 2000 Apr 1;164(7):3596-9. doi: 10.4049/jimmunol.164.7.3596. J Immunol. 2000. PMID: 10725715
-
Decreased indoleamine 2,3-dioxygenase expression in dendritic cells and role of indoleamine 2,3-dioxygenase-expressing dendritic cells in immune thrombocytopenia.Ann Hematol. 2012 Oct;91(10):1623-31. doi: 10.1007/s00277-012-1451-0. Epub 2012 Apr 13. Ann Hematol. 2012. PMID: 22526360
-
T cell regulatory plasmacytoid dendritic cells expressing indoleamine 2,3 dioxygenase.Handb Exp Pharmacol. 2009;(188):165-96. doi: 10.1007/978-3-540-71029-5_8. Handb Exp Pharmacol. 2009. PMID: 19031026 Free PMC article. Review.
-
Tryptophan catabolism in IDO+ plasmacytoid dendritic cells.Curr Drug Metab. 2007 Apr;8(3):209-16. doi: 10.2174/138920007780362581. Curr Drug Metab. 2007. PMID: 17430109 Review.
Cited by
-
Development of a DNA aptamer targeting IDO1 with anti-tumor effects.iScience. 2023 Jul 13;26(8):107367. doi: 10.1016/j.isci.2023.107367. eCollection 2023 Aug 18. iScience. 2023. PMID: 37520707 Free PMC article.
-
Vitamin D3 Priming of Dendritic Cells Shifts Human Neutrophil-Dependent Th17 Cell Development to Regulatory T Cells.Front Immunol. 2022 Jul 7;13:872665. doi: 10.3389/fimmu.2022.872665. eCollection 2022. Front Immunol. 2022. PMID: 35874744 Free PMC article.
-
Targeting the Kynurenine Pathway: A Novel Approach in Tumor Therapy.Expert Rev Mol Med. 2025 Mar 5;27:1-33. doi: 10.1017/erm.2025.5. Online ahead of print. Expert Rev Mol Med. 2025. PMID: 40040508 Free PMC article. Review.
-
Opposing tumor-cell-intrinsic and -extrinsic roles of the IRF1 transcription factor in antitumor immunity.Cell Rep. 2024 Jun 25;43(6):114289. doi: 10.1016/j.celrep.2024.114289. Epub 2024 Jun 2. Cell Rep. 2024. PMID: 38833371 Free PMC article.
-
Therapeutic Liposomal Vaccines for Dendritic Cell Activation or Tolerance.Front Immunol. 2021 May 13;12:674048. doi: 10.3389/fimmu.2021.674048. eCollection 2021. Front Immunol. 2021. PMID: 34054859 Free PMC article. Review.
References
-
- Munn, D.H. , Sharma, M.D. , Baban, B. , Harding, H.P. , Zhang, Y. , Ron, D. , and Mellor, A.L. , GCN2 kinase in T cells mediates proliferative arrest and anergy induction in response to indoleamine 2,3‐dioxygenase. Immunity. 2005. 22: 633–642. - PubMed
-
- Fallarino, F. , Grohmann, U. , You, S. , McGrath, B.C. , Cavener, D.R. , Vacca, C. , Orabona, C. , et al., The combined effects of tryptophan starvation and tryptophan catabolites down‐regulate T cell receptor zeta‐chain and induce a regulatory phenotype in naive T cells. J. Immunol. 2006. 176: 6752–6761. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

