Protecting P-glycoprotein at the blood-brain barrier from degradation in an Alzheimer's disease mouse model
- PMID: 33676539
- PMCID: PMC7937299
- DOI: 10.1186/s12987-021-00245-4
Protecting P-glycoprotein at the blood-brain barrier from degradation in an Alzheimer's disease mouse model
Abstract
Background: Failure to clear Aβ from the brain is partly responsible for Aβ brain accumulation in Alzheimer's disease (AD). A critical protein for clearing Aβ across the blood-brain barrier is the efflux transporter P-glycoprotein (P-gp). In AD, P-gp levels are reduced, which contributes to impaired Aβ brain clearance. However, the mechanism responsible for decreased P-gp levels is poorly understood and there are no strategies available to protect P-gp. We previously demonstrated in isolated brain capillaries ex vivo that human Aβ40 (hAβ40) triggers P-gp degradation by activating the ubiquitin-proteasome pathway. In this pathway, hAβ40 initiates P-gp ubiquitination, leading to internalization and proteasomal degradation of P-gp, which then results in decreased P-gp protein expression and transport activity levels. Here, we extend this line of research and present results from an in vivo study using a transgenic mouse model of AD (human amyloid precursor protein (hAPP)-overexpressing mice; Tg2576).
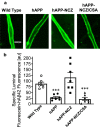
Methods: In our study, hAPP mice were treated with vehicle, nocodazole (NCZ, microtubule inhibitor to block P-gp internalization), or a combination of NCZ and the P-gp inhibitor cyclosporin A (CSA). We determined P-gp protein expression and transport activity levels in isolated mouse brain capillaries and Aβ levels in plasma and brain tissue.
Results: Treating hAPP mice with 5 mg/kg NCZ for 14 days increased P-gp levels to levels found in WT mice. Consistent with this, P-gp-mediated hAβ42 transport in brain capillaries was increased in NCZ-treated hAPP mice compared to untreated hAPP mice. Importantly, NCZ treatment significantly lowered hAβ40 and hAβ42 brain levels in hAPP mice, whereas hAβ40 and hAβ42 levels in plasma remained unchanged.
Conclusions: These findings provide in vivo evidence that microtubule inhibition maintains P-gp protein expression and transport activity levels, which in turn helps to lower hAβ brain levels in hAPP mice. Thus, protecting P-gp at the blood-brain barrier may provide a novel therapeutic strategy for AD and other Aβ-based pathologies.
Keywords: Alzheimer’s disease; Amyloid beta; Blood–brain barrier; Brain capillaries; P-glycoprotein; Ubiquitin-proteasome system.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures






References
-
- Gravina SA, Ho L, Eckman CB, Long KE, Otvos L, Jr, Younkin LH, et al. Amyloid beta protein (A beta) in Alzheimer’s disease brain. Biochemical and immunocytochemical analysis with antibodies specific for forms ending at A beta 40 or A beta 42(43) J Biol Chem. 1995;270(13):7013–6. doi: 10.1074/jbc.270.13.7013. - DOI - PubMed
-
- Frangione Zlokovic B. Transport-clearance hypothesis for Alzheimer’s disease and potential therapeutic implications. Landes Biosci. 2003;54:114–22.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

