TFEB: A Emerging Regulator in Lipid Homeostasis for Atherosclerosis
- PMID: 33679452
- PMCID: PMC7925399
- DOI: 10.3389/fphys.2021.639920
TFEB: A Emerging Regulator in Lipid Homeostasis for Atherosclerosis
Abstract
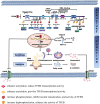
Atherosclerosis, predominantly characterized by the disturbance of lipid homeostasis, has become the main causation of various cardiovascular diseases. Therefore, there is an urgent requirement to explore efficacious targets that act as lipid modulators for atherosclerosis. Transcription factor EB (TFEB), whose activity depends on post-translational modifications, such as phosphorylation, acetylation, SUMOylation, ubiquitination, etc., is significant for normal cell physiology. Recently, increasing evidence implicates a role of TFEB in lipid homeostasis, via its functionality of promoting lipid degradation and efflux through mediating lipophagy, lipolysis, and lipid metabolism-related genes. Furthermore, a regulatory effect on lipid transporters and lipid mediators by TFEB is emerging. Notably, TFEB makes a possible therapeutic target of atherosclerosis by regulating lipid metabolism. This review recapitulates the update and current advances on TFEB mediating lipid metabolism to focus on two intracellular activities: a) how cells perceive external stimuli and initiate transcription programs to modulate TFEB function, and b) how TFEB restores lipid homeostasis in the atherosclerotic process. In-depth research is warranted to develop potent agents against TFEB to alleviate or reverse the progression of atherosclerosis.
Keywords: TFEB; atherosclerosis; lipid homeostasis; lipid mediators; lipid transporters; lipophagy; post-translational modifications.
Copyright © 2021 Li, Wang, Wang, Li and Yang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Hypericin-mediated sonodynamic therapy induces autophagy and decreases lipids in THP-1 macrophage by promoting ROS-dependent nuclear translocation of TFEB.Cell Death Dis. 2016 Dec 22;7(12):e2527. doi: 10.1038/cddis.2016.433. Cell Death Dis. 2016. PMID: 28005078 Free PMC article.
-
The regulation of TFEB in lipid homeostasis of non-alcoholic fatty liver disease: Molecular mechanism and promising therapeutic targets.Life Sci. 2020 Apr 1;246:117418. doi: 10.1016/j.lfs.2020.117418. Epub 2020 Feb 10. Life Sci. 2020. PMID: 32057899 Review.
-
The Role and Regulatory Mechanism of Transcription Factor EB in Health and Diseases.Front Cell Dev Biol. 2021 Aug 13;9:667750. doi: 10.3389/fcell.2021.667750. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34490237 Free PMC article. Review.
-
Transcription factor EB and TFE3: new metabolic coordinators mediating adaptive responses to exercise in skeletal muscle?Am J Physiol Endocrinol Metab. 2020 Oct 1;319(4):E763-E768. doi: 10.1152/ajpendo.00339.2020. Epub 2020 Aug 24. Am J Physiol Endocrinol Metab. 2020. PMID: 32830550 Review.
-
Formononetin alleviates hepatic steatosis by facilitating TFEB-mediated lysosome biogenesis and lipophagy.J Nutr Biochem. 2019 Nov;73:108214. doi: 10.1016/j.jnutbio.2019.07.005. Epub 2019 Jul 25. J Nutr Biochem. 2019. PMID: 31520816
Cited by
-
Association between TFEB gene polymorphism, gene-environment interaction, and fatty liver disease: A case-control study in China.Sci Prog. 2021 Jul-Sep;104(3):368504211043766. doi: 10.1177/00368504211043766. Sci Prog. 2021. PMID: 34581652 Free PMC article.
-
The transcription factors Tfeb and Tfe3 are required for survival and embryonic development of pancreas and liver in zebrafish.PLoS Genet. 2025 Jun 27;21(6):e1011754. doi: 10.1371/journal.pgen.1011754. eCollection 2025 Jun. PLoS Genet. 2025. PMID: 40577485 Free PMC article.
-
Deregulation of mTORC1-TFEB axis in human iPSC model of GBA1-associated Parkinson's disease.Front Neurosci. 2023 Jun 2;17:1152503. doi: 10.3389/fnins.2023.1152503. eCollection 2023. Front Neurosci. 2023. PMID: 37332877 Free PMC article.
-
FGF21 via mitochondrial lipid oxidation promotes physiological vascularization in a mouse model of Phase I ROP.Angiogenesis. 2023 Aug;26(3):409-421. doi: 10.1007/s10456-023-09872-x. Epub 2023 Mar 21. Angiogenesis. 2023. PMID: 36943533 Free PMC article.
-
The Role of Punicalagin and Its Metabolites in Atherosclerosis and Risk Factors Associated with the Disease.Int J Mol Sci. 2023 May 9;24(10):8476. doi: 10.3390/ijms24108476. Int J Mol Sci. 2023. PMID: 37239823 Free PMC article. Review.
References
-
- Argani P., Laé M., Hutchinson B., Reuter V. E., Collins M. H., Perentesis J., et al. (2005). Renal carcinomas with the t(6;11)(p21;q12): clinicopathologic features and demonstration of the specific alpha-TFEB gene fusion by immunohistochemistry, RT-PCR, and DNA PCR. Am. J. Surg. Pathol. 29 230–240. 10.1097/01.pas.0000146007.54092.37 - DOI - PubMed
-
- Argani P., Reuter V. E., Zhang L., Sung Y. S., Ning Y., Epstein J. I., et al. (2016). TFEB-amplified renal cell carcinomas: an aggressive molecular subset demonstrating variable melanocytic marker expression and morphologic heterogeneity. Am. J. Surg. Pathol. 40 1484–1495. 10.1097/pas.0000000000000720 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

