HPLC-MS/MS-Mediated Analysis of the Pharmacokinetics, Bioavailability, and Tissue Distribution of Schisandrol B in Rats
- PMID: 33679983
- PMCID: PMC7929678
- DOI: 10.1155/2021/8862291
HPLC-MS/MS-Mediated Analysis of the Pharmacokinetics, Bioavailability, and Tissue Distribution of Schisandrol B in Rats
Abstract
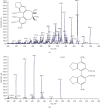
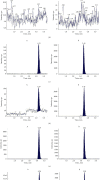
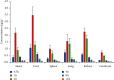
Schisandrol B, a lignan isolated from dried Schisandra chinensis fruits, has been shown to exhibit hepatoprotective, cardioprotective, renoprotective, and memory-enhancing properties. This study sought to design a sensitive and efficient HPLC-MS/MS approach to measuring Schisandrol B levels in rat plasma and tissues in order to assess the pharmacokinetics, oral bioavailability, and tissue distributions of this compound in vivo. For this analysis, bifendate was chosen as an internal standard (IS). A liquid-liquid extraction (LLE) approach was employed for the preparation of samples that were subsequently separated with an Agilent ZORBAX Eclipse XDB-C18 (4.6 × 150 mm, 5 μm) column with an isocratic mobile phase consisting of methanol and water containing 5 mM ammonium acetate and 0.1% formic acid (90 : 10, v/v). A linear calibration curve was obtained over the 5-2000 ng/mL and 1-1000 ng/mL ranges for plasma samples and tissue homogenates, respectively. This established method was then successfully applied to investigate the pharmacokinetics, oral bioavailability, and tissue distributions of Schisandrol B in Sprague-Dawley (SD) rats that were intravenously administered 2 mg/kg of Schisandrol B monomer, intragastrically administered Schisandrol B monomer (10 mg/kg), or intragastrically administered 6 mL/kg SCE (equivalent to 15 mg/kg Schisandrol B monomer). The oral absolute bioavailability of Schisandrol B following intragastric Schisandrol B monomer and SCE administration was approximately 18.73% and 68.12%, respectively. Tissue distribution studies revealed that Schisandrol B was distributed throughout several tested tissues, with particular accumulation in the liver and kidneys. Our data represent a valuable foundation for future studies of the pharmacologic and biological characteristics of Schisandrol B.
Copyright © 2021 Dahu Liang et al.
Conflict of interest statement
All authors of this manuscript declare no conflicts of interest.
Figures

References
LinkOut - more resources
Full Text Sources
Other Literature Sources

