General Control of Amino Acid Synthesis 5-Like 1-Mediated Acetylation of Manganese Superoxide Dismutase Regulates Oxidative Stress in Diabetic Kidney Disease
- PMID: 33680286
- PMCID: PMC7906818
- DOI: 10.1155/2021/6691226
General Control of Amino Acid Synthesis 5-Like 1-Mediated Acetylation of Manganese Superoxide Dismutase Regulates Oxidative Stress in Diabetic Kidney Disease
Abstract
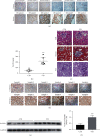
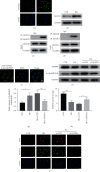
Diabetic kidney disease (DKD) is the major cause of end-stage renal disease (ESRD). In the past few decades, there has been a large amount of evidence to highlight the pivotal role of oxidative stress in the development and progression of DKD. However, the detailed molecular mechanisms are not fully elucidated. A new sight has been established that the mitochondrial acetyltransferase GCN5L1 participates in cellular redox homeostasis maintenance in DKD. Firstly, we found that the expression of GCN5L1 is significantly elevated both in human and mouse kidney tissues with DKD and in hyperglycemic renal tubular epithelial cells (TECs), while deletion of GCN5L1 could effectively ameliorate oxidative stress-induced renal injury in DKD. Furthermore, deletion of GCN5L1 could reduce MnSOD acetylation on lysine 68 and activate its activity, thereby scavenging excessive ROS and relieving oxidative stress-induced renal inflammation and fibrosis. In general, GCN5L1-mediated acetylation of MnSOD exacerbated oxidative stress-induced renal injury, suggesting that GCN5L1 might be a potential intervention target in DKD.
Copyright © 2021 Tingting Lv et al.
Conflict of interest statement
The authors declare that there is no conflict of interest regarding the publication of this paper.
Figures




Similar articles
-
Cardiomyocyte-specific deletion of GCN5L1 in mice restricts mitochondrial protein hyperacetylation in response to a high fat diet.Sci Rep. 2020 Jun 30;10(1):10665. doi: 10.1038/s41598-020-67812-x. Sci Rep. 2020. PMID: 32606301 Free PMC article.
-
GCN5L1 controls renal lipotoxicity through regulating acetylation of fatty acid oxidation enzymes.J Physiol Biochem. 2019 Nov;75(4):597-606. doi: 10.1007/s13105-019-00711-6. Epub 2019 Nov 23. J Physiol Biochem. 2019. PMID: 31760589
-
Elevation of ISG15 promotes diabetic kidney disease by modulating renal tubular epithelial cell pyroptosis.Clin Transl Med. 2025 Jun;15(6):e70337. doi: 10.1002/ctm2.70337. Clin Transl Med. 2025. PMID: 40462493 Free PMC article.
-
Oxidative stress as a culprit in diabetic kidney disease.Life Sci. 2023 Jun 1;322:121661. doi: 10.1016/j.lfs.2023.121661. Epub 2023 Apr 5. Life Sci. 2023. PMID: 37028547 Review.
-
The emerging roles of GCN5L1 in mitochondrial and vacuolar organelle biology.Biochim Biophys Acta Gene Regul Mech. 2021 Feb;1864(2):194598. doi: 10.1016/j.bbagrm.2020.194598. Epub 2020 Jun 26. Biochim Biophys Acta Gene Regul Mech. 2021. PMID: 32599084 Free PMC article. Review.
Cited by
-
GCN5L1 regulates pulmonary surfactant production by modulating lamellar body biogenesis and trafficking in mouse alveolar epithelial cells.Cell Mol Biol Lett. 2023 Nov 7;28(1):90. doi: 10.1186/s11658-023-00506-0. Cell Mol Biol Lett. 2023. PMID: 37936104 Free PMC article.
-
Effects of antioxidants on diabetic kidney diseases: mechanistic interpretations and clinical assessment.Chin Med. 2023 Jan 9;18(1):3. doi: 10.1186/s13020-022-00700-w. Chin Med. 2023. PMID: 36624538 Free PMC article. Review.
-
MRPL12-ANT3 interaction involves in acute kidney injury via regulating MPTP of tubular epithelial cells.iScience. 2023 Apr 14;26(5):106656. doi: 10.1016/j.isci.2023.106656. eCollection 2023 May 19. iScience. 2023. PMID: 37182101 Free PMC article.
-
Manganese Superoxide Dismutase: Structure, Function, and Implications in Human Disease.Antioxidants (Basel). 2025 Jul 10;14(7):848. doi: 10.3390/antiox14070848. Antioxidants (Basel). 2025. PMID: 40722952 Free PMC article. Review.
-
GPER-dependent estrogen signaling increases cardiac GCN5L1 expression.Am J Physiol Heart Circ Physiol. 2022 May 1;322(5):H762-H768. doi: 10.1152/ajpheart.00024.2022. Epub 2022 Mar 4. Am J Physiol Heart Circ Physiol. 2022. PMID: 35245133 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

