Ferrotoxicity and Its Amelioration by Calcitriol in Cultured Renal Cells
- PMID: 33680716
- PMCID: PMC7925041
- DOI: 10.1155/2021/6634429
Ferrotoxicity and Its Amelioration by Calcitriol in Cultured Renal Cells
Abstract
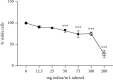
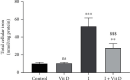
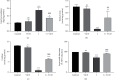
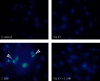
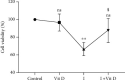
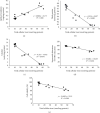
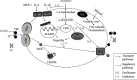
Globally, acute kidney injury (AKI) is associated with significant mortality and an enormous economic burden. Whereas iron is essential for metabolically active renal cells, it has the potential to cause renal cytotoxicity by promoting Fenton chemistry-based oxidative stress involving lipid peroxidation. In addition, 1,25-dihydroxyvitamin D3 (calcitriol), the active form of vitamin D, is reported to have an antioxidative role. In this study, we intended to demonstrate the impact of vitamin D on iron-mediated oxidant stress and cytotoxicity of Vero cells exposed to iohexol, a low osmolar iodine-containing contrast media in vitro. Cultured Vero cells were pretreated with 1,25-dihydroxyvitamin D3 dissolved in absolute ethanol (0.05%, 2.0 mM) at a dose of 1 mM for 6 hours. Subsequently, iohexol was added at a concentration of 100 mg iodine per mL and incubated for 3 hours. Total cellular iron content was analysed by a flame atomic absorption spectrophotometer at 372 nm. Lipid peroxidation was determined by TBARS (thiobarbituric acid reactive species) assay. Antioxidants including total thiol content were assessed by Ellman's method, catalase by colorimetric method, and superoxide dismutase (SOD) by nitroblue tetrazolium assay. The cells were stained with DAPI (4',6-diamidino-2-phenylindole), and the cytotoxicity was evaluated by viability assay (MTT assay). The results indicated that iohexol exposure caused a significant increase of the total iron content in Vero cells. A concomitant increase of lipid peroxidation and decrease of total thiol protein levels, catalase, and superoxide dismutase activity were observed along with decreased cell viability in comparison with the controls. Furthermore, these changes were significantly reversed when the cells were pretreated with vitamin D prior to incubation with iohexol. Our findings of this in vitro model of iohexol-induced renotoxicity lend further support to the nephrotoxic potential of iron and underpin the possible clinical utility of vitamin D for the treatment and prevention of AKI.
Copyright © 2021 Chandrashekar Annamalai et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Figures

References
-
- Lakhmir S., Chawla, on behalf of the Acute Disease Quality Initiative Workgroup 16, Bellomo R., et al. Acute kidney disease and renal recovery: consensus report of the acute disease quality initiative (ADQI) 16 workgroup. Nature Reviews Nephrology. 2017;13(4):241–257. doi: 10.1038/nrneph.2017.2. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

