Functional and Structural Correlates of Impaired Enrichment-Mediated Adult Hippocampal Neurogenesis in a Mouse Model of Prenatal Alcohol Exposure
- PMID: 33680847
- PMCID: PMC7902980
- DOI: 10.3233/BPL-200112
Functional and Structural Correlates of Impaired Enrichment-Mediated Adult Hippocampal Neurogenesis in a Mouse Model of Prenatal Alcohol Exposure
Abstract
Background: Fetal alcohol spectrum disorders (FASDs) are associated with a wide range of cognitive deficiencies.
Objective: We previously found that gestational exposure to moderate levels of alcohol in mice throughout the 1st-2nd human trimester-equivalents for brain development results in profound impairment of the hippocampal neurogenic response to enriched environment (EE) in adulthood, without altering baseline neurogenesis rate under standard housing (SH). However, the functional and structural consequences of impaired EE-mediated neurogenesis in the context of prenatal alcohol exposure (PAE) have not been determined.
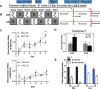
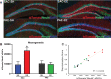
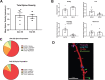
Results: Here, we demonstrate that PAE-EE mice display impaired performance on a neurogenesis-dependent pattern discrimination task, broadened behavioral activation of the dentate gyrus, as assessed by expression of the immediate early gene, c-Fos, and impaired dendritic branching of adult-generated dentate granule cells (aDGCs).
Conclusions: These studies further underscore the impact of moderate gestational alcohol exposure on adult hippocampal plasticity and support adult hippocampal neurogenesis as a potential therapeutic target to remediate certain neurological outcomes in FASD.
Keywords: Pattern discrimination learning; dentate gyrus; fetal alcohol spectrum disorders; immediate-early genes.
© 2020 – IOS Press and the authors. All rights reserved.
Conflict of interest statement
The authors have no conflicts of interest to report.
Figures


Similar articles
-
Resistance of Postnatal Hippocampal Neurogenesis to Alcohol Toxicity in a Third Trimester-Equivalent Mouse Model of Gestational Alcohol Exposure.Alcohol Clin Exp Res. 2019 Dec;43(12):2504-2513. doi: 10.1111/acer.14207. Epub 2019 Oct 20. Alcohol Clin Exp Res. 2019. PMID: 31573091 Free PMC article.
-
Prenatal alcohol exposure alters synaptic activity of adult hippocampal dentate granule cells under conditions of enriched environment.Hippocampus. 2016 Aug;26(8):1078-87. doi: 10.1002/hipo.22588. Epub 2016 Apr 7. Hippocampus. 2016. PMID: 27009742 Free PMC article.
-
Fate analysis of adult hippocampal progenitors in a murine model of fetal alcohol spectrum disorder (FASD).PLoS One. 2013 Sep 11;8(9):e73788. doi: 10.1371/journal.pone.0073788. eCollection 2013. PLoS One. 2013. PMID: 24040071 Free PMC article.
-
Moderate fetal alcohol exposure impairs the neurogenic response to an enriched environment in adult mice.Alcohol Clin Exp Res. 2005 Nov;29(11):2053-62. doi: 10.1097/01.alc.0000187037.02670.59. Alcohol Clin Exp Res. 2005. PMID: 16340464
-
Persistent impairment of hippocampal neurogenesis in young adult rats following early postnatal alcohol exposure.Alcohol Clin Exp Res. 2007 Dec;31(12):2073-82. doi: 10.1111/j.1530-0277.2007.00528.x. Epub 2007 Oct 19. Alcohol Clin Exp Res. 2007. PMID: 17949464
Cited by
-
Adult Hippocampal Neurogenesis as a Therapeutic Target in Fetal Alcohol Spectrum Disorder.Adv Exp Med Biol. 2025;1473:93-109. doi: 10.1007/978-3-031-81908-7_5. Adv Exp Med Biol. 2025. PMID: 40128476 Review.
-
Persistent myelin abnormalities in a third trimester-equivalent mouse model of fetal alcohol spectrum disorder.Alcohol Clin Exp Res. 2022 Jan;46(1):77-86. doi: 10.1111/acer.14752. Epub 2021 Dec 15. Alcohol Clin Exp Res. 2022. PMID: 34825395 Free PMC article.
-
Comparative Analysis of Test Tube and Volumetric Drinking Monitor Methods in Voluntary Ethanol Consumption in Female Mice for Prenatal Alcohol Exposure.bioRxiv [Preprint]. 2025 Jun 12:2025.06.10.658718. doi: 10.1101/2025.06.10.658718. bioRxiv. 2025. PMID: 40661392 Free PMC article. Preprint.
References
-
- Greenmyer JR, Popova S, Klug MG, Burd L. Fetal alcohol spectrum disorder: a systematic review of the cost of and savings from prevention in the United States and Canada. Addiction. 2020;115(3):409–17. - PubMed
-
- Tan CH, Denny CH, Cheal NE, Sniezek JE, Kanny D. Alcohol use and binge drinking among women of childbearing age - United States, 2011–2013. MMWR Morbidity and mortality weekly report. 2015;64(37):1042–6. - PubMed
-
- Kodituwakku PW, Kodituwakku EL. From research to practice: an integrative framework for the development of interventions for children with fetal alcohol spectrum disorders. Neuropsychology Review. 2011;21(2):204–23. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
