Inhibition of astrocyte hemichannel improves recovery from spinal cord injury
- PMID: 33682795
- PMCID: PMC8021110
- DOI: 10.1172/jci.insight.134611
Inhibition of astrocyte hemichannel improves recovery from spinal cord injury
Abstract
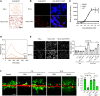
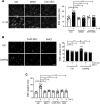
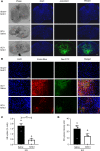
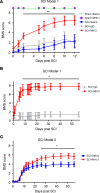
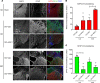
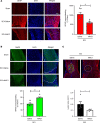
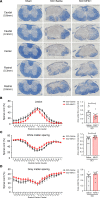
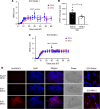
Spinal cord injury (SCI) causes severe disability, and the current inability to restore function to the damaged spinal cord leads to lasting detrimental consequences to patients. One strategy to reduce SCI morbidity involves limiting the spread of secondary damage after injury. Previous studies have shown that connexin 43 (Cx43), a gap junction protein richly expressed in spinal cord astrocytes, is a potential mediator of secondary damage. Here, we developed a specific inhibitory antibody, mouse-human chimeric MHC1 antibody (MHC1), that inhibited Cx43 hemichannels, but not gap junctions, and reduced secondary damage in 2 incomplete SCI mouse models. MHC1 inhibited the activation of Cx43 hemichannels in both primary spinal astrocytes and astrocytes in situ. In both SCI mouse models, administration of MHC1 after SCI significantly improved hind limb locomotion function. Remarkably, a single administration of MHC1 30 minutes after injury improved the recovery up to 8 weeks post-SCI. Moreover, MHC1 treatment decreased gliosis and lesion sizes, increased white and gray matter sparing, and improved neuronal survival. Together, these results suggest that inhibition of Cx43 hemichannel function after traumatic SCI reduces secondary damage, limits perilesional gliosis, and improves functional recovery. By targeting hemichannels specifically with an antibody, this study provides a potentially new, innovative therapeutic approach in treating SCI.
Keywords: Neurological disorders; Therapeutics.
Conflict of interest statement
Figures
Similar articles
-
Connexin43 and astrocytic gap junctions in the rat spinal cord after acute compression injury.J Comp Neurol. 1997 Jun 2;382(2):199-214. J Comp Neurol. 1997. PMID: 9183689
-
Astrocytic CX43 hemichannels and gap junctions play a crucial role in development of chronic neuropathic pain following spinal cord injury.Glia. 2012 Nov;60(11):1660-70. doi: 10.1002/glia.22384. Epub 2012 Aug 1. Glia. 2012. PMID: 22951907 Free PMC article.
-
Astrocyte sigma-1 receptors modulate connexin 43 expression leading to the induction of below-level mechanical allodynia in spinal cord injured mice.Neuropharmacology. 2016 Dec;111:34-46. doi: 10.1016/j.neuropharm.2016.08.027. Epub 2016 Aug 24. Neuropharmacology. 2016. PMID: 27567941
-
Role of connexins in spinal cord injury: An update.Clin Neurol Neurosurg. 2020 Oct;197:106102. doi: 10.1016/j.clineuro.2020.106102. Epub 2020 Jul 21. Clin Neurol Neurosurg. 2020. PMID: 32717564 Review.
-
Connexin and pannexin hemichannels in inflammatory responses of glia and neurons.Brain Res. 2012 Dec 3;1487:3-15. doi: 10.1016/j.brainres.2012.08.042. Epub 2012 Sep 10. Brain Res. 2012. PMID: 22975435 Free PMC article. Review.
Cited by
-
Fascin-1 limits myosin activity in microglia to control mechanical characterization of the injured spinal cord.J Neuroinflammation. 2024 Apr 10;21(1):88. doi: 10.1186/s12974-024-03089-5. J Neuroinflammation. 2024. PMID: 38600569 Free PMC article.
-
Activation of connexin hemichannels enhances mechanosensitivity and anabolism in disused and aged bone.JCI Insight. 2024 Dec 6;9(23):e177557. doi: 10.1172/jci.insight.177557. JCI Insight. 2024. PMID: 39641271 Free PMC article.
-
Boldine modulates glial transcription and functional recovery in a murine model of contusion spinal cord injury.Front Cell Neurosci. 2023 Jun 21;17:1163436. doi: 10.3389/fncel.2023.1163436. eCollection 2023. Front Cell Neurosci. 2023. PMID: 37416508 Free PMC article.
-
Identifying signature genes and their associations with immune cell infiltration in spinal cord injury.IBRO Neurosci Rep. 2024 Sep 21;17:320-328. doi: 10.1016/j.ibneur.2024.09.002. eCollection 2024 Dec. IBRO Neurosci Rep. 2024. PMID: 39430218 Free PMC article.
-
Gap Junctions or Hemichannel-Dependent and Independent Roles of Connexins in Fibrosis, Epithelial-Mesenchymal Transitions, and Wound Healing.Biomolecules. 2023 Dec 14;13(12):1796. doi: 10.3390/biom13121796. Biomolecules. 2023. PMID: 38136665 Free PMC article. Review.
References
-
- O’Carroll SJ, et al. Connexin 43 mimetic peptides reduce swelling, astrogliosis, and neuronal cell death after spinal cord injury. Cell Commun Adhes. 2008;15(1):27–42. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

