SARS-CoV-2 suppresses anticoagulant and fibrinolytic gene expression in the lung
- PMID: 33683204
- PMCID: PMC8049742
- DOI: 10.7554/eLife.64330
SARS-CoV-2 suppresses anticoagulant and fibrinolytic gene expression in the lung
Abstract
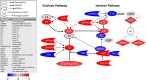
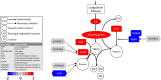
Extensive fibrin deposition in the lungs and altered levels of circulating blood coagulation proteins in COVID-19 patients imply local derangement of pathways that limit fibrin formation and/or promote its clearance. We examined transcriptional profiles of bronchoalveolar lavage fluid (BALF) samples to identify molecular mechanisms underlying these coagulopathies. mRNA levels for regulators of the kallikrein-kinin (C1-inhibitor), coagulation (thrombomodulin, endothelial protein C receptor), and fibrinolytic (urokinase and urokinase receptor) pathways were significantly reduced in COVID-19 patients. While transcripts for several coagulation proteins were increased, those encoding tissue factor, the protein that initiates coagulation and whose expression is frequently increased in inflammatory disorders, were not increased in BALF from COVID-19 patients. Our analysis implicates enhanced propagation of coagulation and decreased fibrinolysis as drivers of the coagulopathy in the lungs of COVID-19 patients.
Keywords: COVID-19; SARS-CoV-2; bronchoalvelolar; coagulation; epidemiology; fibrinolysis; global health; human; medicine.
© 2021, Mast et al.
Conflict of interest statement
AM receives research funding from Novo Nordisk and has received honoraria for serving on Novo Nordisk advisory boards. AW receives research funding from Takeda and Bristol Myers Squibb, DG receives research funding from Bayer and has received honoraria for serving on Anthos, Bristol-Myers Squibb, Ionis and Janssen advisory boards. MG, CA, JM, BA, DJ No competing interests declared
Figures
Comment in
-
Comment on 'SARS-CoV-2 suppresses anticoagulant and fibrinolytic gene expression in the lung'.Elife. 2022 Jan 11;11:e74268. doi: 10.7554/eLife.74268. Elife. 2022. PMID: 35014954 Free PMC article.
References
-
- Adachi T, Chong JM, Nakajima N, Sano M, Yamazaki J, Miyamoto I, Nishioka H, Akita H, Sato Y, Kataoka M, Katano H, Tobiume M, Sekizuka T, Itokawa K, Kuroda M, Suzuki T. Clinicopathologic and immunohistochemical findings from autopsy of patient with COVID-19, japan. Emerging Infectious Diseases. 2020;26:2157–2161. doi: 10.3201/eid2609.201353. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- HL143403/NH/NIH HHS/United States
- HL068835/NH/NIH HHS/United States
- 3RF1AG053303-01S2/AG/NIA NIH HHS/United States
- HL126974/NH/NIH HHS/United States
- U01 HL143403/HL/NHLBI NIH HHS/United States
- U24 HL148865/NH/NIH HHS/United States
- R35 HL140025/HL/NHLBI NIH HHS/United States
- R01 HL068835/HL/NHLBI NIH HHS/United States
- U24 HL148865/HL/NHLBI NIH HHS/United States
- RF1 AG053303/AG/NIA NIH HHS/United States
- P30 DK078392/DK/NIDDK NIH HHS/United States
- HL140025/NH/NIH HHS/United States
- R01 HL126974/HL/NHLBI NIH HHS/United States
- U24 HL148/NH/NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

