Palmitic acid promotes resistin-induced insulin resistance and inflammation in SH-SY5Y human neuroblastoma
- PMID: 33686181
- PMCID: PMC7940652
- DOI: 10.1038/s41598-021-85018-7
Palmitic acid promotes resistin-induced insulin resistance and inflammation in SH-SY5Y human neuroblastoma
Erratum in
-
Publisher Correction: Palmitic acid promotes resistin‑induced insulin resistance and inflammation in SH‑SY5Y human neuroblastoma.Sci Rep. 2021 Jun 15;11(1):12935. doi: 10.1038/s41598-021-92151-w. Sci Rep. 2021. PMID: 34131233 Free PMC article. No abstract available.
Abstract
Saturated fatty acids such as palmitic acid promote inflammation and insulin resistance in peripheral tissues, contrasting with the protective action of polyunsaturated fatty acids such docosahexaenoic acid. Palmitic acid effects have been in part attributed to its potential action through Toll-like receptor 4. Beside, resistin, an adipokine, also promotes inflammation and insulin resistance via TLR4. In the brain, palmitic acid and resistin trigger neuroinflammation and insulin resistance, but their link at the neuronal level is unknown. Using human SH-SY5Yneuroblastoma cell line we show that palmitic acid treatment impaired insulin-dependent Akt and Erk phosphorylation whereas DHA preserved insulin action. Palmitic acid up-regulated TLR4 as well as pro-inflammatory cytokines IL6 and TNFα contrasting with DHA effect. Similarly to palmitic acid, resistin treatment induced the up-regulation of IL6 and TNFα as well as NFκB activation. Importantly, palmitic acid potentiated the resistin-dependent NFkB activation whereas DHA abolished it. The recruitment of TLR4 to membrane lipid rafts was increased by palmitic acid treatment; this is concomitant with the augmentation of resistin-induced TLR4/MYD88/TIRAP complex formation mandatory for TLR4 signaling. In conclusion, palmitic acid increased TLR4 expression promoting resistin signaling through TLR4 up-regulation and its recruitment to membrane lipid rafts.
Conflict of interest statement
The authors declare no competing interests.
Figures
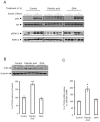
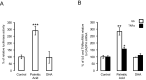

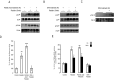


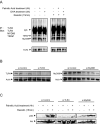
References
-
- Ilievski V, Cho Y, Katwala P, Rodriguez H, Tulowiecka M, Kurian D, Leoni L, Christman JW, Unterman TG, Watanabe K. TLR4 expression by liver resident cells mediates the development of glucose intolerance and insulin resistance in experimental periodontitis. PLoS ONE. 2015;10(8):e0136502. doi: 10.1371/journal.pone.0136502. - DOI - PMC - PubMed
-
- Zheng C, Yang Q, Xu C, Shou P, Cao J, Jiang M, Chen Q, Cao G, Han Y, Li F, Cao W, Zhang L, Shi Y, Wang Y. CD11b regulates obesity-induced insulin resistance via limiting activation and proliferation of adipose tissue macrophages. Proc. Natl. Acad. Sci. USA. 2015;112:E7239–E7248. doi: 10.1073/pnas.1500396113. - DOI - PMC - PubMed
-
- Kain V, Kapadia B, Viswakarma N, Seshadri S, Prajapati B, Jena PK, Meda CLT, Subramanian M, Suraj SK, Kumar ST, Babu PP, Thimmapaya B, Reddy JK, Parsa KVL, Misra P. Co-activator binding protein PIMT mediates TNFa induced insulin resistance in skeletal muscle via the transcriptional down-regulation of MEF2A and Glut4. Sci. Rep. 2015;5:15197. doi: 10.1038/srep15197. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

