Effects of Ih and TASK-like shunting current on dendritic impedance in layer 5 pyramidal-tract neurons
- PMID: 33689489
- PMCID: PMC8282219
- DOI: 10.1152/jn.00015.2021
Effects of Ih and TASK-like shunting current on dendritic impedance in layer 5 pyramidal-tract neurons
Abstract
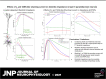
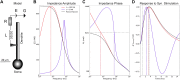
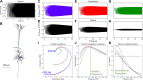
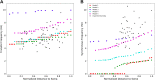
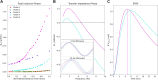
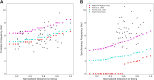
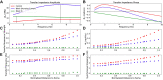
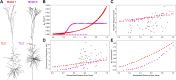
Pyramidal neurons in neocortex have complex input-output relationships that depend on their morphologies, ion channel distributions, and the nature of their inputs, but which cannot be replicated by simple integrate-and-fire models. The impedance properties of their dendritic arbors, such as resonance and phase shift, shape neuronal responses to synaptic inputs and provide intraneuronal functional maps reflecting their intrinsic dynamics and excitability. Experimental studies of dendritic impedance have shown that neocortical pyramidal tract neurons exhibit distance-dependent changes in resonance and impedance phase with respect to the soma. We, therefore, investigated how well several biophysically detailed multicompartment models of neocortical layer 5 pyramidal tract neurons reproduce the location-dependent impedance profiles observed experimentally. Each model tested here exhibited location-dependent impedance profiles, but most captured either the observed impedance amplitude or phase, not both. The only model that captured features from both incorporates hyperpolarization-activated cyclic nucleotide-gated (HCN) channels and a shunting current, such as that produced by Twik-related acid-sensitive K+ (TASK) channels. TASK-like channel density in this model was proportional to local HCN channel density. We found that although this shunting current alone is insufficient to produce resonance or realistic phase response, it modulates all features of dendritic impedance, including resonance frequencies, resonance strength, synchronous frequencies, and total inductive phase. We also explored how the interaction of HCN channel current (Ih) and a TASK-like shunting current shape synaptic potentials and produce degeneracy in dendritic impedance profiles, wherein different combinations of Ih and shunting current can produce the same impedance profile.NEW & NOTEWORTHY We simulated chirp current stimulation in the apical dendrites of 5 biophysically detailed multicompartment models of neocortical pyramidal tract neurons and found that a combination of HCN channels and TASK-like channels produced the best fit to experimental measurements of dendritic impedance. We then explored how HCN and TASK-like channels can shape the dendritic impedance as well as the voltage response to synaptic currents.
Keywords: Twik-related acid-sensitive K+(TASK) channels; h-current (Ih); impedance; pyramidal tract neurons; resonance.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
- R01 DC012947/DC/NIDCD NIH HHS/United States
- U24 EB028998/EB/NIBIB NIH HHS/United States
- R01MH086638/HHS | National Institutes of Health (NIH)
- R01EB022903/HHS | National Institutes of Health (NIH)
- INTERNET2 E-CAS 1904444/National Science Foundation
- R01DC012947-06A1/HHS | NIH | National Institute on Deafness and Other Communication Disorders (NIDCD)
- U01EB017695/HHS | National Institutes of Health (NIH)
- NYS DOH01-C32250GG-3450000/New York State Department of Health
- W911NF-19-1-0402/Army Research Office
- U01 EB017695/EB/NIBIB NIH HHS/United States
- R01 MH086638/MH/NIMH NIH HHS/United States
- P50 MH109429/MH/NIMH NIH HHS/United States
- R01 EB022903/EB/NIBIB NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

