Identification of an N'-Nitrosonornicotine-Specific Deoxyadenosine Adduct in Rat Liver and Lung DNA
- PMID: 33705110
- PMCID: PMC8942342
- DOI: 10.1021/acs.chemrestox.1c00013
Identification of an N'-Nitrosonornicotine-Specific Deoxyadenosine Adduct in Rat Liver and Lung DNA
Abstract
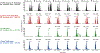
The tobacco-specific nitrosamines N'-nitrosonornicotine (NNN) and 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK) are considered to be two of the most important carcinogens in unburned tobacco and its smoke. They readily cause tumors in laboratory animals and are classified as "carcinogenic to humans" by the International Agency for Research on Cancer. DNA adduct formation by these two carcinogens is believed to play a critical role in tobacco carcinogenesis. Among all the DNA adducts formed by NNN and NNK, 2'-deoxyadenosine (dAdo)-derived adducts have not been fully characterized. In the study reported here, we characterized the formation of N6-[4-(3-pyridyl)-4-oxo-1-butyl]-2'-deoxyadenosine (N6-POB-dAdo) and its reduced form N6-PHB-dAdo formed by NNN 2'-hydroxylation in rat liver and lung DNA. More importantly, we characterized a new dAdo adduct N6-[4-hydroxy-1-(pyridine-3-yl)butyl]-2'-deoxyadenosine (N6-HPB-dAdo) formed after NaBH3CN or NaBH4 reduction both in vitro in calf thymus DNA reacted with 5'-acetoxy-N'-nitrosonornicotine and in vivo in rat liver and lung upon treatment with NNN. This adduct was specifically formed by NNN 5'-hydroxylation. Chemical standards of N6-HPB-dAdo and the corresponding isotopically labeled internal standard [pyridine-d4]N6-HPB-dAdo were synthesized using a four-step method. Both NMR and high-resolution mass spectrometry data agreed well with the proposed structure of N6-HPB-dAdo. The new adduct coeluted with the synthesized internal standard under various LC conditions. Its product ion patterns of MS2 and MS3 transitions were also consistent with the proposed fragmentation patterns. Chromatographic resolution of the two diastereomers of N6-HPB-dAdo was successfully achieved. Quantitation suggested a dose-dependent response of the levels of this new adduct in the liver and lung of rats treated with NNN. However, its level was lower than that of 2-[2-(3-pyridyl)-N-pyrrolidinyl]-2'-deoxyinosine, a previously reported dGuo adduct that is also formed from NNN 5'-hydroxylation. The identification of N6-HPB-dAdo in this study leads to new insights pertinent to the mechanism of carcinogenesis by NNN and to the development of biomarkers of NNN metabolic activation.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
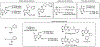

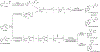
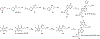
References
-
- Islami F, Goding Sauer A, Miller KD, Siegel RL, Fedewa SA, Jacobs EJ, McCullough ML, Patel AV, Ma J, et al. (2018) Proportion and number of cancer cases and deaths attributable to potentially modifiable risk factors in the United States. CA Cancer J Clin 68, 31–54. - PubMed
-
- Hecht SS (1998) Biochemistry, biology, and carcinogenicity of tobacco-specific N-nitrosamines. Chem Res Toxicol 11, 559–603. - PubMed
-
- Hecht SS (2020) Metabolism and DNA adduct formation of carcinogenic tobacco-specific nitrosamines found in smokeless tobacco products, In Smokeless Tobacco Products: Characteristics, Usage, Health Effects, and Regulatory Implications (Pickworth WB, Ed.) pp 151–166, Elsevier, Amsterdam.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

