Misdiagnosed thrombocytopenia in children and adolescents: analysis of the Pediatric and Adult Registry on Chronic ITP
- PMID: 33710335
- PMCID: PMC7993109
- DOI: 10.1182/bloodadvances.2020003004
Misdiagnosed thrombocytopenia in children and adolescents: analysis of the Pediatric and Adult Registry on Chronic ITP
Abstract
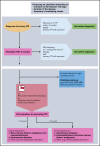
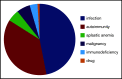
Primary immune thrombocytopenia (ITP) in children is a diagnosis of exclusion, but cases of secondary ITP and nonimmune thrombocytopenia (non-IT) are generally difficult to recognize in a timely fashion. We describe a pediatric population with a revised diagnosis of secondary ITP or non-IT within 24 months of follow-up. Data were extracted from the Pediatric and Adult Registry on Chronic ITP, an international multicenter registry collecting data prospectively in patients with newly diagnosed primary ITP. Between 2004 and 2019, a total of 3974 children aged 3 months to 16 years were included. Secondary ITP and non-IT were reported in 113 patients (63 female subjects). Infectious (n = 53) and autoimmune (n = 42) diseases were identified as the main causes, with median ages at diagnosis of 3.2 years (interquartile range: 1.2; 6.7 years) and 12.4 years (interquartile range: 7.6; 13.7 years), respectively. Other causes included malignancies, aplastic anemia, immunodeficiency, and drug use. Patients with malignancy and aplastic anemia had significantly higher initial platelet counts (37 and 52 × 109/L) than did those with infection or autoimmune diseases (12 and 13 × 109/L). Characteristics of patients with secondary ITP due to infection were similar to those of children with primary ITP at first presentation, indicating similar mechanisms. Significant differences were found for age, sex, comorbidities, initial bleeding, sustained need for treatment, and disease persistence for the remaining noninfectious group compared with primary ITP. Based on our findings, we propose a diagnostic algorithm that may serve as a basis for further discussion and prospective trials.
© 2021 by The American Society of Hematology.
Conflict of interest statement
Conflict-of-interest disclosure: A.S. reports advisory board membership for Novartis and research funds from Amgen. M.M. serves as a consultant for Novartis, Amgen, and Rigel. T.K. reports research funds from Novartis and Amgen; and advisory board membership for UCB Biosciences. S.H. reports advisory board membership for Novartis. M.G.S. reports advisory board membership for Jazz Pharmaceuticals; honoraria from Novartis; and travel grants from Shire and Amgen. The remaining authors declare no competing financial interests.
Figures


References
-
- Rodeghiero F, Stasi R, Gernsheimer T, et al. . Standardization of terminology, definitions and outcome criteria in immune thrombocytopenic purpura of adults and children: report from an international working group. Blood. 2009;113(11):2386-2393. - PubMed
-
- Liebman H. Other immune thrombocytopenias. Semin Hematol. 2007;44(4suppl 5):S24-S34. - PubMed
-
- Bussel JB. Therapeutic approaches to secondary immune thrombocytopenic purpura. Semin Hematol. 2009;46(1suppl 2):S44-S58. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

