Extension study of the safety and efficacy of CLS-TA for treatment of macular oedema associated with non-infectious uveitis (MAGNOLIA)
- PMID: 33712478
- PMCID: PMC9340030
- DOI: 10.1136/bjophthalmol-2020-317560
Extension study of the safety and efficacy of CLS-TA for treatment of macular oedema associated with non-infectious uveitis (MAGNOLIA)
Abstract
Purpose: To assess the extended efficacy and safety of suprachoroidal triamcinolone acetonide injectable suspension (CLS-TA) among patients with macular oedema (ME) secondary to non-infectious uveitis (NIU).
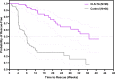
Methods: Patients with uveitic ME were treated with suprachoroidal CLS-TA at baseline and week 12 of the Efficacy and Safety of Suprachoroidal CLS-TA for Macular Edema Secondary to Noninfectious Uveitis: Phase 3 Randomized Trial (PEACHTREE) study. Time to rescue was evaluated over 24 additional weeks for MAGNOLIA. Safety data, visual acuity and retinal central subfield thickness (CST) reduction were also evaluated. Of the 53 eligible patients (46 CLS-TA and 7 control), 33 patients were enrolled (28 CLS-TA and 5 control).
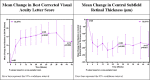
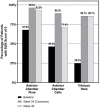
Results: Over the entire 48-week period for PEACHTREE and MAGNOLIA, the median time to rescue therapy was 257 days versus 55.5 days for the CLS-TA and sham-control arms, respectively. Of 28 CLS-TA treated patients who participated in MAGNOLIA, 14 (50%) did not require rescue therapy through approximately 9 months after the second treatment. Among CLS-TA patients not requiring rescue, there was a mean gain of 12.1 letters and mean CST reduction of 174.5 µm at week 48. No serious adverse events related to study treatment were observed.
Conclusion: Approximately 50% of patients did not require additional treatment for up to 9 months following the last CLS-TA administration.
Keywords: macula; retina.
© Author(s) (or their employer(s)) 2022. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: RNK reports receiving consulting fees from Allergan, Genentech and Regeneron. He also reports receiving grant support from Allergan, Chengdu Kanghong, Clearside Biomedical, Roche and Santen. PM reports receiving consulting fees from Eyepoint, Alimera and Allergan. She also reports receiving grant support from Clearside, Gilead, Santen and NEI. SY reports receiving consulting fees from Clearside Biomedical and Santen. He also reports receiving grant support from Bayer. ES reports receiving consulting fees from Clearside Biomedical, Abbvie, Eyegate, Eyepoint and Gilead. He also reports receiving grant support from Aldeyra. MRB reports receiving consulting fees from Allegro, Allergan, Alimera, Bausch and Lomb, Genentech, Novartis and Regenxbio. EU reports receiving consulting fees from Abbvie, Novartis, Eyepoint, Regeneron, GlaxoSmithKline and Bausch and Lomb. He also reports receiving grant support from Novartis and Roche/Genentech. CRH reports receiving consulting fees from Clearside Biomedical. MS reports receiving grant support from Clearside Biomedical. RCW has nothing to disclose. TC and BK are employees of Clearside Biomedical and hold stock in Clearside Biomedical.
Figures
Similar articles
-
Suprachoroidal Space Triamcinolone Acetonide: A Review in Uveitic Macular Edema.Drugs. 2022 Sep;82(13):1403-1410. doi: 10.1007/s40265-022-01763-7. Epub 2022 Aug 26. Drugs. 2022. PMID: 36018461 Free PMC article. Review.
-
Suprachoroidal triamcinolone acetonide versus rescue therapies for the treatment of uveitic macular oedema: A post hoc analysis of PEACHTREE.Clin Exp Ophthalmol. 2022 Jan;50(1):23-30. doi: 10.1111/ceo.14024. Epub 2021 Dec 27. Clin Exp Ophthalmol. 2022. PMID: 34741564 Free PMC article. Clinical Trial.
-
Efficacy and Safety of Suprachoroidal CLS-TA for Macular Edema Secondary to Noninfectious Uveitis: Phase 3 Randomized Trial.Ophthalmology. 2020 Jul;127(7):948-955. doi: 10.1016/j.ophtha.2020.01.006. Epub 2020 Jan 10. Ophthalmology. 2020. PMID: 32173113 Clinical Trial.
-
Suprachoroidal CLS-TA with and without Systemic Corticosteroid and/or Steroid-Sparing Therapy: A Post-Hoc Analysis of the Phase 3 PEACHTREE Clinical Trial.Ocul Immunol Inflamm. 2023 Oct;31(8):1579-1586. doi: 10.1080/09273948.2021.1954199. Epub 2021 Aug 18. Ocul Immunol Inflamm. 2023. PMID: 34406900 Free PMC article.
-
Microinjection via the suprachoroidal space: a review of a novel mode of administration.Am J Manag Care. 2022 Nov;28(13 Suppl):S243-S252. doi: 10.37765/ajmc.2022.89270. Am J Manag Care. 2022. PMID: 36395492 Review.
Cited by
-
Early adoption of triamcinolone acetonide suprachoroidal injection for uveitic macular edema: a physician survey.BMC Res Notes. 2024 Oct 23;17(1):317. doi: 10.1186/s13104-024-06969-4. BMC Res Notes. 2024. PMID: 39444009 Free PMC article.
-
Suprachoroidal Space Triamcinolone Acetonide: A Review in Uveitic Macular Edema.Drugs. 2022 Sep;82(13):1403-1410. doi: 10.1007/s40265-022-01763-7. Epub 2022 Aug 26. Drugs. 2022. PMID: 36018461 Free PMC article. Review.
-
Drug Delivery via the Suprachoroidal Space for the Treatment of Retinal Diseases.Pharmaceutics. 2021 Jun 26;13(7):967. doi: 10.3390/pharmaceutics13070967. Pharmaceutics. 2021. PMID: 34206925 Free PMC article. Review.
-
System Analysis Based on Lipid-Metabolism-Related Genes Identifies AGT as a Novel Therapy Target for Gastric Cancer with Neoadjuvant Chemotherapy.Pharmaceutics. 2023 Mar 2;15(3):810. doi: 10.3390/pharmaceutics15030810. Pharmaceutics. 2023. PMID: 36986671 Free PMC article.
-
Suprachoroidal Injection of Triamcinolone Acetonide Suspension: Ocular Pharmacokinetics and Distribution in Rabbits Demonstrates High and Durable Levels in the Chorioretina.J Ocul Pharmacol Ther. 2022 Jul-Aug;38(6):459-467. doi: 10.1089/jop.2021.0090. Epub 2022 Apr 11. J Ocul Pharmacol Ther. 2022. PMID: 35404132 Free PMC article.
References
-
- Thorne JE, Sugar EA, Holbrook JT, et al. . Periocular triamcinolone vs. intravitreal triamcinolone vs. intravitreal dexamethasone implant for the treatment of uveitic macular edema: the periocular vs. intravitreal corticosteroids for uveitic macular edema (point) trial. Ophthalmology 2019;126:283–95. 10.1016/j.ophtha.2018.08.021 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
