Between-trial heterogeneity in ARDS research
- PMID: 33713156
- PMCID: PMC7955690
- DOI: 10.1007/s00134-021-06370-w
Between-trial heterogeneity in ARDS research
Abstract
Purpose: Most randomized controlled trials (RCTs) in patients with acute respiratory distress syndrome (ARDS) revealed indeterminate or conflicting study results. We aimed to systematically evaluate between-trial heterogeneity in reporting standards and trial outcome.
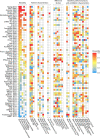
Methods: A systematic review of RCTs published between 2000 and 2019 was performed including adult ARDS patients receiving lung-protective ventilation. A random-effects meta-regression model was applied to quantify heterogeneity (non-random variability) and to evaluate trial and patient characteristics as sources of heterogeneity.
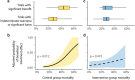
Results: In total, 67 RCTs were included. The 28-day control-group mortality rate ranged from 10 to 67% with large non-random heterogeneity (I2 = 88%, p < 0.0001). Reported baseline patient characteristics explained some of the outcome heterogeneity, but only six trials (9%) reported all four independently predictive variables (mean age, mean lung injury score, mean plateau pressure and mean arterial pH). The 28-day control group mortality adjusted for patient characteristics (i.e. the residual heterogeneity) ranged from 18 to 45%. Trials with significant benefit in the primary outcome reported a higher control group mortality than trials with an indeterminate outcome or harm (mean 28-day control group mortality: 44% vs. 28%; p = 0.001).
Conclusion: Among ARDS RCTs in the lung-protective ventilation era, there was large variability in the description of baseline characteristics and significant unexplainable heterogeneity in 28-day control group mortality. These findings signify problems with the generalizability of ARDS research and underline the urgent need for standardized reporting of trial and baseline characteristics.
Keywords: ARDS; Critical Care Research; Heterogeneity.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Figures

References
-
- Bellani G, Laffey JG, Pham T, Fan E, Brochard L, Esteban A, et al. Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 Countries. JAMA. 2016;315(8):788–800. - PubMed
-
- Rubenfeld GD, Caldwell E, Peabody E, Weaver J, Martin DP, Neff M, et al. Incidence and outcomes of acute lung injury. N Engl J Med. 2005;353(16):1685–1693. - PubMed
-
- Villar J, Blanco J, Añón JM, Santos-Bouza A, Blanch L, Ambrós A, et al. The ALIEN study: incidence and outcome of acute respiratory distress syndrome in the era of lung protective ventilation. Intensive Care Med. 2011;37(12):1932–1941. - PubMed
-
- Fan E, Del Sorbo L, Goligher EC, Hodgson CL, Munshi L, Walkey AJ, et al. An Official American Thoracic Society/European Society of Intensive Care Medicine/Society Of Critical Care Medicine clinical practice guideline: mechanical ventilation in adult patients with acute respiratory distress syndrome. Am J Respir Crit Care Med. 2017;195(9):1253–1263. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

