Stem Cell-Based Clinical Trials for Diabetes Mellitus
- PMID: 33716982
- PMCID: PMC7953062
- DOI: 10.3389/fendo.2021.631463
Stem Cell-Based Clinical Trials for Diabetes Mellitus
Abstract
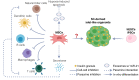
Since its introduction more than twenty years ago, intraportal allogeneic cadaveric islet transplantation has been shown to be a promising therapy for patients with Type I Diabetes (T1D). Despite its positive outcome, the impact of islet transplantation has been limited due to a number of confounding issues, including the limited availability of cadaveric islets, the typically lifelong dependence of immunosuppressive drugs, and the lack of coverage of transplant costs by health insurance companies in some countries. Despite improvements in the immunosuppressive regimen, the number of required islets remains high, with two or more donors per patient often needed. Insulin independence is typically achieved upon islet transplantation, but on average just 25% of patients do not require exogenous insulin injections five years after. For these reasons, implementation of islet transplantation has been restricted almost exclusively to patients with brittle T1D who cannot avoid hypoglycemic events despite optimized insulin therapy. To improve C-peptide levels in patients with both T1 and T2 Diabetes, numerous clinical trials have explored the efficacy of mesenchymal stem cells (MSCs), both as supporting cells to protect existing β cells, and as source for newly generated β cells. Transplantation of MSCs is found to be effective for T2D patients, but its efficacy in T1D is controversial, as the ability of MSCs to differentiate into functional β cells in vitro is poor, and transdifferentiation in vivo does not seem to occur. Instead, to address limitations related to supply, human embryonic stem cell (hESC)-derived β cells are being explored as surrogates for cadaveric islets. Transplantation of allogeneic hESC-derived insulin-producing organoids has recently entered Phase I and Phase II clinical trials. Stem cell replacement therapies overcome the barrier of finite availability, but they still face immune rejection. Immune protective strategies, including coupling hESC-derived insulin-producing organoids with macroencapsulation devices and microencapsulation technologies, are being tested to balance the necessity of immune protection with the need for vascularization. Here, we compare the diverse human stem cell approaches and outcomes of recently completed and ongoing clinical trials, and discuss innovative strategies developed to overcome the most significant challenges remaining for transplanting stem cell-derived β cells.
Keywords: clinical trial (CT); encapsulation; islets; stem cells; transplantation; type 1 diabetes (T1D); type 2 diabetes (T2D).
Copyright © 2021 de Klerk and Hebrok.
Conflict of interest statement
MH owns stocks/stock options in Viacyte, Encellin, Thymmune, and Minutia. MH also serves as SAB member to 1351 Thymmune and Encellin, and is co-Founder, SAB and Board member for 1352 EndoCrine and Minutia. The remaining author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Bretzel RG, Hering BJ, Schultz AO, Geier C, Federlin K. International islet transplant registry report. In: Lanza RP, Chick WL, editors. Yearbook of Cell and Tissue Transplantation 1996–1997. Dordrecht: Springer Netherlands; (1996). p. 153–60. 10.1007/978-94-009-0165-0_15 - DOI
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

