Identification of Immune-Related Therapeutically Relevant Biomarkers in Breast Cancer and Breast Cancer Stem Cells by Transcriptome-Wide Analysis: A Clinical Prospective Study
- PMID: 33718103
- PMCID: PMC7945036
- DOI: 10.3389/fonc.2020.554138
Identification of Immune-Related Therapeutically Relevant Biomarkers in Breast Cancer and Breast Cancer Stem Cells by Transcriptome-Wide Analysis: A Clinical Prospective Study
Abstract
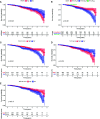
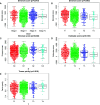
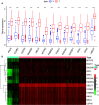
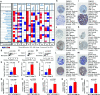
Cancer stem cells (CSCs) represent a subset of tumor cells that are responsible for recurrence and metastasis of tumors. These cells are resistant to radiotherapy and chemotherapy. Immunotherapeutic strategies that target CSCs specifically have provided initial results; however, the mechanism of action of these strategies is unclear. The data were requested from The Cancer Genome Atlas and Genotype-Tissue Expression, followed with the survival analysis and weighted gene co-expression network analysis to detect survival and stemness related genes. Patients were divided into three groups based on their immune status by applying single sample GSEA (ssGSEA) with proven dependability by ESTIMATE analysis. The filtered key genes were analyzed using oncomine, GEPIA, HPA, qRT-PCR, and functional analysis. Patients in a group with a higher stemness and a lower immune infiltration showed a worse overall survival probability, stemness and immune infiltration characteristics of breast cancer progressed in a non-linear fashion. Thirteen key genes related to stemness and immunity were identified and the functional analysis indicated their crucial roles in cell proliferation and immune escape strategies. The qRT-PCR results showed that the expression of PIMREG and MTFR2 differed in different stages of patients. Our study revealed a promising potential for CSC-target immunotherapy in the early stage of cancer and a probable value for PIMREG and MTFR2 as biomarkers and targets for immunotherapy.
Keywords: MTFR2; PIMREG; breast cancer; cancer stem cell; tumor immune infiltration.
Copyright © 2021 Wang, Liu, Liu, Wang, Tai, Yin and Tan.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures










Similar articles
-
Stemness Related Genes Revealed by Network Analysis Associated With Tumor Immune Microenvironment and the Clinical Outcome in Lung Adenocarcinoma.Front Genet. 2020 Sep 16;11:549213. doi: 10.3389/fgene.2020.549213. eCollection 2020. Front Genet. 2020. PMID: 33193623 Free PMC article.
-
Identification of key genes controlling breast cancer stem cell characteristics via stemness indices analysis.J Transl Med. 2020 Feb 12;18(1):74. doi: 10.1186/s12967-020-02260-9. J Transl Med. 2020. PMID: 32050983 Free PMC article.
-
Pan-Cancer Analysis of PIMREG as a Biomarker for the Prognostic and Immunological Role.Front Genet. 2021 Sep 14;12:687778. doi: 10.3389/fgene.2021.687778. eCollection 2021. Front Genet. 2021. PMID: 34594356 Free PMC article.
-
Concise Review: Targeting Cancer Stem Cells Using Immunologic Approaches.Stem Cells. 2015 Jul;33(7):2085-92. doi: 10.1002/stem.2039. Epub 2015 May 13. Stem Cells. 2015. PMID: 25873269 Free PMC article. Review.
-
Evolving Strategies for Therapeutically Targeting Cancer Stem Cells.Adv Cancer Res. 2016;131:159-91. doi: 10.1016/bs.acr.2016.04.003. Epub 2016 Jun 3. Adv Cancer Res. 2016. PMID: 27451127 Review.
Cited by
-
Investigating combined hypoxia and stemness indices for prognostic transcripts in gastric cancer: Machine learning and network analysis approaches.Biochem Biophys Rep. 2024 Dec 19;41:101897. doi: 10.1016/j.bbrep.2024.101897. eCollection 2025 Mar. Biochem Biophys Rep. 2024. PMID: 39807391 Free PMC article.
-
Regulatory Functions of microRNAs in Cancer Stem Cells: Mechanism, Facts, and Perspectives.Cells. 2025 Jul 14;14(14):1073. doi: 10.3390/cells14141073. Cells. 2025. PMID: 40710326 Free PMC article. Review.
-
TPD52 as a Potential Prognostic Biomarker and its Correlation with Immune Infiltrates in Uterine Corpus Endometrial Carcinoma: Bioinformatic Analysis and Experimental Verification.Recent Pat Anticancer Drug Discov. 2025;20(1):71-88. doi: 10.2174/0115748928267447231107101539. Recent Pat Anticancer Drug Discov. 2025. PMID: 38305309
-
ZP3 Expression in Pancreatic Adenocarcinoma: Its Implications for the Prognosis and Therapy.Protein Pept Lett. 2025;32(2):124-138. doi: 10.2174/0109298665350171241204153202. Protein Pept Lett. 2025. PMID: 39791146
-
ADAMTSL2 is a potential prognostic biomarker and immunotherapeutic target for colorectal cancer: Bioinformatic analysis and experimental verification.PLoS One. 2024 May 30;19(5):e0303909. doi: 10.1371/journal.pone.0303909. eCollection 2024. PLoS One. 2024. PMID: 38814950 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources

