Functional Role of Class III Myosins in Hair Cells
- PMID: 33718386
- PMCID: PMC7947357
- DOI: 10.3389/fcell.2021.643856
Functional Role of Class III Myosins in Hair Cells
Abstract
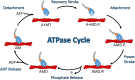
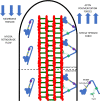
Cytoskeletal motors produce force and motion using the energy from ATP hydrolysis and function in a variety of mechanical roles in cells including muscle contraction, cargo transport, and cell division. Actin-based myosin motors have been shown to play crucial roles in the development and function of the stereocilia of auditory and vestibular inner ear hair cells. Hair cells can contain hundreds of stereocilia, which rely on myosin motors to elongate, organize, and stabilize their structure. Mutations in many stereocilia-associated myosins have been shown to cause hearing loss in both humans and animal models suggesting that each myosin isoform has a specific function in these unique parallel actin bundle-based protrusions. Here we review what is known about the classes of myosins that function in the stereocilia, with a special focus on class III myosins that harbor point mutations associated with delayed onset hearing loss. Much has been learned about the role of the two class III myosin isoforms, MYO3A and MYO3B, in maintaining the precise stereocilia lengths required for normal hearing. We propose a model for how class III myosins play a key role in regulating stereocilia lengths and demonstrate how their motor and regulatory properties are particularly well suited for this function. We conclude that ongoing studies on class III myosins and other stereocilia-associated myosins are extremely important and may lead to novel therapeutic strategies for the treatment of hearing loss due to stereocilia degeneration.
Keywords: ATPase; MYO3A; MYO3B; actin; force generation; myosin; stereocilia.
Copyright © 2021 Cirilo, Gunther and Yengo.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Pathophysiology of human hearing loss associated with variants in myosins.Front Physiol. 2024 Mar 18;15:1374901. doi: 10.3389/fphys.2024.1374901. eCollection 2024. Front Physiol. 2024. PMID: 38562617 Free PMC article. Review.
-
The dynamics of actin protrusions can be controlled by tip-localized myosin motors.J Biol Chem. 2024 Jan;300(1):105516. doi: 10.1016/j.jbc.2023.105516. Epub 2023 Nov 30. J Biol Chem. 2024. PMID: 38042485 Free PMC article.
-
Class III myosins shape the auditory hair bundles by limiting microvilli and stereocilia growth.J Cell Biol. 2016 Jan 18;212(2):231-44. doi: 10.1083/jcb.201509017. Epub 2016 Jan 11. J Cell Biol. 2016. PMID: 26754646 Free PMC article.
-
Stereocilia-staircase spacing is influenced by myosin III motors and their cargos espin-1 and espin-like.Nat Commun. 2016 Mar 1;7:10833. doi: 10.1038/ncomms10833. Nat Commun. 2016. PMID: 26926603 Free PMC article.
-
Stereocilia morphogenesis and maintenance through regulation of actin stability.Semin Cell Dev Biol. 2017 May;65:88-95. doi: 10.1016/j.semcdb.2016.08.017. Epub 2016 Aug 23. Semin Cell Dev Biol. 2017. PMID: 27565685 Free PMC article. Review.
Cited by
-
Pathophysiology of human hearing loss associated with variants in myosins.Front Physiol. 2024 Mar 18;15:1374901. doi: 10.3389/fphys.2024.1374901. eCollection 2024. Front Physiol. 2024. PMID: 38562617 Free PMC article. Review.
-
MYO3B promotes cancer progression in endometrial cancer by mediating the calcium ion-RhoA/ROCK1 signaling pathway.J Cancer Res Clin Oncol. 2024 Sep 19;150(9):424. doi: 10.1007/s00432-024-05940-x. J Cancer Res Clin Oncol. 2024. PMID: 39297944 Free PMC article.
-
Hearing Function: Identification of New Candidate Genes Further Explaining the Complexity of This Sensory Ability.Genes (Basel). 2021 Aug 10;12(8):1228. doi: 10.3390/genes12081228. Genes (Basel). 2021. PMID: 34440402 Free PMC article.
-
Deafness mutation in the MYO3A motor domain impairs actin protrusion elongation mechanism.Mol Biol Cell. 2022 Jan 1;33(1):ar5. doi: 10.1091/mbc.E21-05-0232. Epub 2021 Nov 17. Mol Biol Cell. 2022. PMID: 34788109 Free PMC article.
-
The dynamics of actin protrusions can be controlled by tip-localized myosin motors.J Biol Chem. 2024 Jan;300(1):105516. doi: 10.1016/j.jbc.2023.105516. Epub 2023 Nov 30. J Biol Chem. 2024. PMID: 38042485 Free PMC article.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

