Fejerlectin, a Lectin-like Peptide from the Skin of Fejervarya limnocharis, Inhibits HIV-1 Entry by Targeting Gp41
- PMID: 33718732
- PMCID: PMC7948434
- DOI: 10.1021/acsomega.1c00033
Fejerlectin, a Lectin-like Peptide from the Skin of Fejervarya limnocharis, Inhibits HIV-1 Entry by Targeting Gp41
Abstract
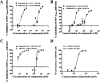
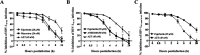
Human immunodeficiency virus type 1 (HIV-1) is mainly transmitted by sexual intercourse, and effective microbicides preventing HIV-1 transmission are still required. Amphibian skin is a rich source of defense peptides with antiviral activity. Here, we characterized a lectin-like peptide, fejerlectin (RLCYMVLPCP), isolated from the skin of the frog Fejervarya limnocharis. Fejerlectin showed significant hemagglutination and d-(+)-galacturonic acid-binding activities. Furthermore, fejerlectin suppressed the early entry of HIV-1 into target cells by binding to the N-terminal heptad repeat of HIV-1 gp41 and preventing 6-HB formation and Env-mediated membrane fusion. Fejerlectin is the smallest lectin-like peptide identified to date and represents a new and promising platform for anti-HIV-1 drug development.
© 2021 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures




Similar articles
-
Conserved Residue Asn-145 in the C-Terminal Heptad Repeat Region of HIV-1 gp41 is Critical for Viral Fusion and Regulates the Antiviral Activity of Fusion Inhibitors.Viruses. 2019 Jul 3;11(7):609. doi: 10.3390/v11070609. Viruses. 2019. PMID: 31277353 Free PMC article.
-
Hydrophobic mutations in buried polar residues enhance HIV-1 gp41 N-terminal heptad repeat-C-terminal heptad repeat interactions and C-peptides' anti-HIV activity.AIDS. 2014 Jun 1;28(9):1251-60. doi: 10.1097/QAD.0000000000000255. AIDS. 2014. PMID: 24625369
-
Creating an Artificial Tail Anchor as a Novel Strategy To Enhance the Potency of Peptide-Based HIV Fusion Inhibitors.J Virol. 2016 Dec 16;91(1):e01445-16. doi: 10.1128/JVI.01445-16. Print 2017 Jan 1. J Virol. 2016. PMID: 27795416 Free PMC article.
-
High throughput screening and characterization of HIV-1 entry inhibitors targeting gp41: theories and techniques.Curr Pharm Des. 2004;10(15):1827-43. doi: 10.2174/1381612043384466. Curr Pharm Des. 2004. PMID: 15180543 Review.
-
Peptide and non-peptide HIV fusion inhibitors.Curr Pharm Des. 2002;8(8):563-80. doi: 10.2174/1381612024607180. Curr Pharm Des. 2002. PMID: 11945159 Review.
Cited by
-
A Review: The Antiviral Activity of Cyclic Peptides.Int J Pept Res Ther. 2023;29(1):7. doi: 10.1007/s10989-022-10478-y. Epub 2022 Dec 1. Int J Pept Res Ther. 2023. PMID: 36471676 Free PMC article. Review.
-
Evaluation of the antiviral activity of new dermaseptin analogs against Zika virus.Biochem Biophys Rep. 2024 Jun 7;39:101747. doi: 10.1016/j.bbrep.2024.101747. eCollection 2024 Sep. Biochem Biophys Rep. 2024. PMID: 38939125 Free PMC article.
-
Cathelicidin-HG Alleviates Sepsis-Induced Platelet Dysfunction by Inhibiting GPVI-Mediated Platelet Activation.Research (Wash D C). 2024 Jun 5;7:0381. doi: 10.34133/research.0381. eCollection 2024. Research (Wash D C). 2024. PMID: 38840901 Free PMC article.
-
[Antibacterial mechanism of Brevinin-2GHk, an antimicrobial peptide from Fejervarya limnocharis skin].Nan Fang Yi Ke Da Xue Xue Bao. 2021 Nov 20;41(11):1657-1663. doi: 10.12122/j.issn.1673-4254.2021.11.09. Nan Fang Yi Ke Da Xue Xue Bao. 2021. PMID: 34916191 Free PMC article. Chinese.
-
Brevinin-2GHk, a Peptide Derived from the Skin of Fejervarya limnocharis, Inhibits Zika Virus Infection by Disrupting Viral Integrity.Viruses. 2021 Nov 28;13(12):2382. doi: 10.3390/v13122382. Viruses. 2021. PMID: 34960651 Free PMC article.
References
-
- Jurado S.; Cano-Muñoz M.; Morel B.; Standoli S.; Santarossa E.; Moog C.; Schmidt S.; Laumond G.; Cámara-Artigas A.; Conejero-Lara F. Structural and Thermodynamic Analysis of HIV-1 Fusion Inhibition Using Small Gp41 Mimetic Proteins. J. Mol. Biol. 2019, 431, 3091–3106. 10.1016/j.jmb.2019.06.022. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

