Strategies Towards Improving Clinical Outcomes of Peptide Receptor Radionuclide Therapy
- PMID: 33721105
- PMCID: PMC7960621
- DOI: 10.1007/s11912-021-01037-7
Strategies Towards Improving Clinical Outcomes of Peptide Receptor Radionuclide Therapy
Abstract
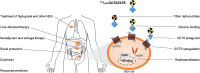
Purpose of review: Peptide receptor radionuclide therapy (PRRT) with [177Lu-DOTA0,Tyr3] octreotate is an effective and safe second- or third-line treatment option for patients with low-grade advanced gastroenteropancreatic (GEP) neuroendocrine neoplasms (NEN). In this review, we will focus on possible extensions of the current use of PRRT and on new approaches which could further improve its treatment efficacy and safety.
Recent findings: Promising results were published regarding PRRT in other NENs, including lung NENs or high-grade NENs, and applying PRRT as neoadjuvant or salvage therapy. Furthermore, a diversity of strategic approaches, including dosimetry, somatostatin receptor antagonists, somatostatin receptor upregulation, radiosensitization, different radionuclides, albumin binding, alternative renal protection, and liver-directed therapy in combination with PRRT, have the potential to improve the outcome of PRRT. Also, novel biomarkers are presented that could predict response to PRRT. Multiple preclinical and early clinical studies have shown encouraging potential to advance the clinical outcome of PRRT in NEN patients. However, at this moment, most of these strategies have not yet reached the clinical setting of randomized phase III trials.
Keywords: Neuroendocrine tumour; Peptide receptor radionuclide therapy.
Conflict of interest statement
Noémie S. Minczeles declares that she has no conflict of interest. Johannes Hofland has received compensation from Advanced Accelerator Applications / Novartis and Ipsen for service on advisory boards, and has received speaker's honorarium from Ipsen. Wouter W. de Herder has received research funding from Ipsen; has received speaker's honoraria from Advanced Accelerator Applications / Novartis, Ipsen, Pfizer, and Sanofi; and has received compensation from Advanced Accelerator Applications / Novartis and Ipsen for service on advisory boards. Tessa Brabander has received compensation from Advanced Accelerator Applications / Novartis for service on an advisory board, and has received speaker's honoraria from Advanced Accelerator Applications / Novartis and Medscape.
Figures
Similar articles
-
Peptide receptor radionuclide therapy in gastroenteropancreatic NEN G3: a multicenter cohort study.Endocr Relat Cancer. 2019 Feb 1;26(2):227-239. doi: 10.1530/ERC-18-0424. Endocr Relat Cancer. 2019. PMID: 30540557
-
Current status of peptide receptor radionuclide therapy in grade 1 and 2 gastroenteropancreatic neuroendocrine tumours.J Neuroendocrinol. 2025 Mar;37(3):e13469. doi: 10.1111/jne.13469. Epub 2024 Nov 20. J Neuroendocrinol. 2025. PMID: 39563515 Free PMC article. Review.
-
Salvage peptide receptor radionuclide therapy with [177Lu-DOTA,Tyr3]octreotate in patients with bronchial and gastroenteropancreatic neuroendocrine tumours.Eur J Nucl Med Mol Imaging. 2019 Mar;46(3):704-717. doi: 10.1007/s00259-018-4158-1. Epub 2018 Sep 28. Eur J Nucl Med Mol Imaging. 2019. PMID: 30267116 Free PMC article.
-
Peptide receptor radionuclide therapy for patients with advanced pancreatic neuroendocrine tumors.Semin Oncol. 2018 Aug;45(4):236-248. doi: 10.1053/j.seminoncol.2018.08.004. Epub 2018 Oct 24. Semin Oncol. 2018. PMID: 30539715 Review.
-
Peptide Receptor Radionuclide Therapy for Advanced Gastroenteropancreatic Neuroendocrine Tumors - from oncology perspective.Nucl Med Rev Cent East Eur. 2018;21(2). doi: 10.5603/NMR.2018.0019. Nucl Med Rev Cent East Eur. 2018. PMID: 29741203 Review.
Cited by
-
Radiation exposure and protection advice after [177Lu]Lu-DOTA-TATE therapy in China.EJNMMI Res. 2024 Nov 28;14(1):119. doi: 10.1186/s13550-024-01185-4. EJNMMI Res. 2024. PMID: 39607652 Free PMC article.
-
Beyond Small Molecules: Antibodies and Peptides for Fibroblast Activation Protein Targeting Radiopharmaceuticals.Pharmaceutics. 2024 Feb 29;16(3):345. doi: 10.3390/pharmaceutics16030345. Pharmaceutics. 2024. PMID: 38543239 Free PMC article. Review.
-
Clinical Utility of 18F-FDG PET in Neuroendocrine Tumors Prior to Peptide Receptor Radionuclide Therapy: A Systematic Review and Meta-Analysis.Cancers (Basel). 2021 Apr 10;13(8):1813. doi: 10.3390/cancers13081813. Cancers (Basel). 2021. PMID: 33920195 Free PMC article. Review.
-
Peptide Radioligands in Cancer Theranostics: Agonists and Antagonists.Pharmaceuticals (Basel). 2023 Apr 30;16(5):674. doi: 10.3390/ph16050674. Pharmaceuticals (Basel). 2023. PMID: 37242457 Free PMC article. Review.
-
Peptide Receptor Radionuclide Therapy.J Clin Endocrinol Metab. 2022 Nov 25;107(12):3199-3208. doi: 10.1210/clinem/dgac574. J Clin Endocrinol Metab. 2022. PMID: 36198028 Free PMC article.
References
-
- Yao JC, Hassan M, Phan A, Dagohoy C, Leary C, Mares JE, Abdalla EK, Fleming JB, Vauthey JN, Rashid A, Evans DB. One hundred years after “carcinoid”: epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. J Clin Oncol. 2008;26(18):3063–3072. doi: 10.1200/JCO.2007.15.4377. - DOI - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous