Double-blind, placebo-controlled randomised clinical trial to evaluate the effect of ASPIRIN discontinuation after left atrial appendage occlusion in atrial fibrillation: protocol of the ASPIRIN LAAO trial
- PMID: 33722871
- PMCID: PMC7970210
- DOI: 10.1136/bmjopen-2020-044695
Double-blind, placebo-controlled randomised clinical trial to evaluate the effect of ASPIRIN discontinuation after left atrial appendage occlusion in atrial fibrillation: protocol of the ASPIRIN LAAO trial
Abstract
Introduction: It is the common clinical practice to prescribe indefinite aspirin for patients with non-valvular atrial fibrillation (NVAF) post left atrial appendage occlusion (LAAO). However, aspirin as a primary prevention strategy for cardiovascular diseases has recently been challenged due to increased risk of bleeding. Therefore, aspirin discontinuation after LAAO in atrial fibrillation (ASPIRIN LAAO) trial is designed to assess the uncertainty about the risks and benefits of discontinuing aspirin therapy at 6 months postimplantation with a Watchman LAAO device in NVAF patients.
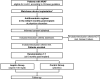
Methods and analysis: The ASPIRIN LAAO study is a prospective, multicentre, randomised, double-blinded, placebo-controlled non-inferiority trial. Patients implanted with a Watchman device within 6 months prior to enrollment and without pre-existing conditions requiring long-term aspirin therapy according to current guidelines are eligible for participating the trial. Subjects will be randomised in a 1:1 allocation ratio to either the Aspirin group (aspirin 100 mg/day) or the control group (placebo) at 6 months postimplantation. A total of 1120 subjects will be enrolled from 12 investigational sites in China. The primary composite endpoint is stroke, systemic embolism, cardiovascular/unexplained death, major bleeding, acute coronary syndrome and coronary or periphery artery disease requiring revascularisation at 24 months. Follow-up visits are scheduled at 6 and 12 months and then every 12 months until 24 months after the last patient recruitment.
Ethics and dissemination: Ethics approval was obtained from the Ethics Committee of Xinhua Hospital, Shanghai, China (reference number XHEC-C-2018-065-5). The protocol is also submitted and approved by the institutional Ethics Committee at each participating centre. Results are expected in 2024 and will be disseminated through peer-reviewed journals and presentations at national and international conferences.
Trial registration number: NCT03821883.
Keywords: adult cardiology; clinical trials; stroke.
© Author(s) (or their employer(s)) 2021. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: None declared.
Figures

References
-
- January CT, Wann LS, Calkins H, et al. 2019 AHA/ACC/HRS focused update of the 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American heart association Task force on clinical practice guidelines and the heart rhythm Society. J Am Coll Cardiol 2019;74:104–32. 10.1016/j.jacc.2019.01.011 - DOI - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
