Phase 2 study of TAS-117, an allosteric akt inhibitor in advanced solid tumors harboring phosphatidylinositol 3-kinase/v-akt murine thymoma viral oncogene homolog gene mutations
- PMID: 33723724
- PMCID: PMC8426297
- DOI: 10.1007/s10637-021-01085-7
Phase 2 study of TAS-117, an allosteric akt inhibitor in advanced solid tumors harboring phosphatidylinositol 3-kinase/v-akt murine thymoma viral oncogene homolog gene mutations
Abstract
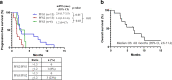
TAS-117 is a potent and selective allosteric pan-v-akt murine thymoma viral oncogene homolog (Akt) inhibitor. We conducted a single-arm single-center phase 2 study of TAS-117 in heavily treated patients with tumors refractory to systemic chemotherapy and harboring phosphatidylinositol 3-kinase (PI3K)/Akt mutations. Patients with gastrointestinal (GI) cancers were orally administered 16 mg TAS-117 daily, and those with non-GI tumors were administered 24 mg on a 4 days on/3 days off schedule. The primary endpoint was overall response rate (ORR). Secondary endpoints included disease control rate (DCR), progression-free survival (PFS), overall survival (OS), PFS ratio, safety, and tolerability. Thirteen patients were enrolled: eight with non-GI (breast, ovarian, endometrial, and non-small cell lung) and five with GI (colon, rectal, gastric, and gallbladder) cancers. Ten patients were treated with TAS-117 after ≥ 4 lines of therapy. Twelve patients showed PIK3 catalytic subunit alpha (PIK3CA) mutations; one harbored an Akt1E17K mutation. The median treatment duration was 1.4 months; the median number of treatment cycles was 2. The ORR was 8 %, and DCR was 23 %. The median PFS and OS were 1.4 and 4.8 months, respectively. Grade 3-4 treatment-related adverse events were anorexia (grade 3, 8 %) and hyperglycemia (grade 3, 8 %; grade 4, 8 %).Grade 3-4 treatment-related adverse events occurred in 27 % of grade 3 anorexia (9 %) and hyperglycemia (grade 3, 8 %; grade 4, 9\%). TAS-117 showed limited antitumor activity and manageable toxicity. Clinical efficacy was observed in patients with ovarian cancer harboring PIK3CA E545K mutations and in patients with breast cancer harboring PIK3CA H1047R and Akt1E17K mutations.Trial registration: This study was retrospectively registered with ClinicalTrial.gov (NCT03017521 on January 11, 2017).
Keywords: Basket trial; PI3K/Akt mutations; Solid tumors; TAS-117.
© 2021. The Author(s).
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Phase II trial of AKT inhibitor MK-2206 in patients with advanced breast cancer who have tumors with PIK3CA or AKT mutations, and/or PTEN loss/PTEN mutation.Breast Cancer Res. 2019 Jul 5;21(1):78. doi: 10.1186/s13058-019-1154-8. Breast Cancer Res. 2019. PMID: 31277699 Free PMC article. Clinical Trial.
-
AKT Inhibition in Solid Tumors With AKT1 Mutations.J Clin Oncol. 2017 Jul 10;35(20):2251-2259. doi: 10.1200/JCO.2017.73.0143. Epub 2017 May 10. J Clin Oncol. 2017. PMID: 28489509 Free PMC article.
-
Phase 2 study of LY3023414 in patients with advanced endometrial cancer harboring activating mutations in the PI3K pathway.Cancer. 2020 Mar 15;126(6):1274-1282. doi: 10.1002/cncr.32677. Epub 2019 Dec 27. Cancer. 2020. PMID: 31880826 Free PMC article. Clinical Trial.
-
"The emerging role of capivasertib in breast cancer".Breast. 2022 Jun;63:157-167. doi: 10.1016/j.breast.2022.03.018. Epub 2022 Apr 1. Breast. 2022. PMID: 35398754 Free PMC article. Review.
-
The PIK3CA gene as a mutated target for cancer therapy.Curr Cancer Drug Targets. 2008 Dec;8(8):733-40. doi: 10.2174/156800908786733504. Curr Cancer Drug Targets. 2008. PMID: 19075596 Free PMC article. Review.
Cited by
-
Basket Trials: Past, Present, and Future.Annu Rev Cancer Biol. 2024 Jun;8(1):59-80. doi: 10.1146/annurev-cancerbio-061421-012927. Epub 2023 Dec 5. Annu Rev Cancer Biol. 2024. PMID: 38938274 Free PMC article.
-
Therapeutic targeting of anoikis resistance in cutaneous melanoma metastasis.Front Cell Dev Biol. 2023 Apr 26;11:1183328. doi: 10.3389/fcell.2023.1183328. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 37181747 Free PMC article. Review.
-
The Growth Modulation Index (GMI) as an Efficacy Outcome in Cancer Clinical Trials: A Scoping Review with Suggested Reporting Guidelines.Curr Oncol Rep. 2025 May;27(5):516-532. doi: 10.1007/s11912-025-01667-1. Epub 2025 Mar 29. Curr Oncol Rep. 2025. PMID: 40156702 Free PMC article.
-
A phase II study of TAS-117 in patients with advanced solid tumors harboring germline PTEN-inactivating mutations.Future Oncol. 2022 Sep;18(30):3377-3387. doi: 10.2217/fon-2022-0305. Epub 2022 Aug 30. Future Oncol. 2022. PMID: 36039910 Free PMC article.
-
Inhibitors of phosphoinositide 3-kinase (PI3K) and phosphoinositide 3-kinase-related protein kinase family (PIKK).J Enzyme Inhib Med Chem. 2023 Dec;38(1):2237209. doi: 10.1080/14756366.2023.2237209. J Enzyme Inhib Med Chem. 2023. PMID: 37489050 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

