RNA-Seq analysis of knocking out the neuroprotective proton-sensitive GPR68 on basal and acute ischemia-induced transcriptome changes and signaling in mouse brain
- PMID: 33724568
- PMCID: PMC7970445
- DOI: 10.1096/fj.202002511R
RNA-Seq analysis of knocking out the neuroprotective proton-sensitive GPR68 on basal and acute ischemia-induced transcriptome changes and signaling in mouse brain
Abstract
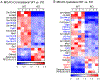
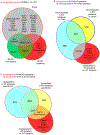
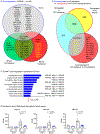
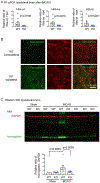
Brain acid signaling plays important roles in both physiological and disease conditions. One key neuronal metabotropic proton receptor in the brain is GPR68, which contributes to hippocampal long-term potentiation (LTP) and mediates neuroprotection in acidotic and ischemic conditions. Here, to gain greater understanding of GPR68 function in the brain, we performed mRNA-Seq analysis in mice. First, we studied sham-operated animals to determine baseline expression. Compared to wild type (WT), GPR68-/- (KO) brain downregulated genes that are enriched in Gene Ontology (GO) terms of misfolding protein binding, response to organic cyclic compounds, and endoplasmic reticulum chaperone complex. Next, we examined the expression profile following transient middle cerebral artery occlusion (tMCAO). tMCAO-upregulated genes cluster to cytokine/chemokine-related functions and immune responses, while tMCAO-downregulated genes cluster to channel activities and synaptic signaling. For proton-sensitive receptors, tMCAO downregulated ASIC1a and upregulated GPR4 and GPR65, but had no effect on ASIC2, PAC, or GPR68. GPR68 deletion did not alter the expression of these proton receptors, either at baseline or after ischemia. Lastly, we performed GeneVenn analysis of differential genes at baseline and post-tMCAO. Ischemia upregulated the expression of three hemoglobin genes, along with H2-Aa, Ppbp, Siglece, and Tagln, in WT but not in KO. Immunostaining showed that tMCAO-induced hemoglobin localized to neurons. Western blot analysis further showed that hemoglobin induction is GPR68-dependent. Together, these data suggest that GPR68 deletion at baseline disrupts chaperone functions and cellular signaling responses and imply a contribution of hemoglobin-mediated antioxidant mechanism to GPR68-dependent neuroprotection in ischemia.
Keywords: GPR68; OGR1; ischemia; neuroprotection; proton receptor; transcriptome.
© 2021 Federation of American Societies for Experimental Biology.
Conflict of interest statement
CONFLICT OF INTEREST STATEMENT
The authors declare no competing interests.
Figures




References
-
- Chesler M, and Kaila K (1992) Modulation of pH by neuronal activity. Trends Neurosci 15, 396–402 - PubMed
-
- Glitsch M (2019) Mechano- and pH-sensing convergence on Ca(2+)-mobilising proteins - A recipe for cancer? Cell Calcium 80, 38–45 - PubMed
-
- Silver IA, and Erecinska M (1992) Ion homeostasis in rat brain in vivo: intra- and extracellular [Ca2+] and [H+] in the hippocampus during recovery from short-term, transient ischemia. J Cereb Blood Flow Metab 12, 759–772 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

