Modulation of Mechanical Stress Mitigates Anti-Dsg3 Antibody-Induced Dissociation of Cell-Cell Adhesion
- PMID: 33724731
- PMCID: PMC7993752
- DOI: 10.1002/adbi.202000159
Modulation of Mechanical Stress Mitigates Anti-Dsg3 Antibody-Induced Dissociation of Cell-Cell Adhesion
Abstract
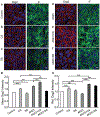
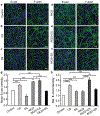
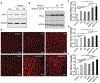
It is becoming increasingly clear that mechanical stress in adhesive junctions plays a significant role in dictating the fate of cell-cell attachment under physiological conditions. Targeted disruption of cell-cell junctions leads to multiple pathological conditions, among them the life-threatening autoimmune blistering disease pemphigus vulgaris (PV). The dissociation of cell-cell junctions by autoantibodies is the hallmark of PV, however, the detailed mechanisms that result in tissue destruction remain unclear. Thus far, research and therapy in PV have focused primarily on immune mechanisms upstream of autoantibody binding, while the biophysical aspects of the cell-cell dissociation process leading to acantholysis are less well studied. In work aimed at illuminating the cellular consequences of autoantibody attachment, it is reported that externally applied mechanical stress mitigates antibody-induced monolayer fragmentation and inhibits p38 MAPK phosphorylation activated by anti-Dsg3 antibody. Further, it is demonstrated that mechanical stress applied externally to cell monolayers enhances cell contractility via RhoA activation and promotes the strengthening of cortical actin, which ultimately mitigates antibody-induced cell-cell dissociation. The study elevates understanding of the mechanism of acantholysis in PV and shifts the paradigm of PV disease development from a focus solely on immune pathways to highlight the key role of physical transformations at the target cell.
Keywords: autoantibodies; cell-cell adhesion; desmosome; mechanical stress; pemphigus.
© 2021 Wiley-VCH GmbH.
Conflict of interest statement
Conflict of Interest
The authors declare no conflict of interest.
Figures

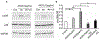
Similar articles
-
Keratin Retraction and Desmoglein3 Internalization Independently Contribute to Autoantibody-Induced Cell Dissociation in Pemphigus Vulgaris.Front Immunol. 2018 Apr 25;9:858. doi: 10.3389/fimmu.2018.00858. eCollection 2018. Front Immunol. 2018. PMID: 29922278 Free PMC article.
-
Adducin is required for desmosomal cohesion in keratinocytes.J Biol Chem. 2014 May 23;289(21):14925-40. doi: 10.1074/jbc.M113.527127. Epub 2014 Apr 7. J Biol Chem. 2014. PMID: 24711455 Free PMC article.
-
Desmoglein endocytosis and desmosome disassembly are coordinated responses to pemphigus autoantibodies.J Biol Chem. 2006 Mar 17;281(11):7623-34. doi: 10.1074/jbc.M512447200. Epub 2005 Dec 23. J Biol Chem. 2006. PMID: 16377623
-
Desmosomal adhesion and pemphigus vulgaris: the first half of the story.Cell Commun Adhes. 2013 Feb;20(1-2):1-10. doi: 10.3109/15419061.2013.763799. Epub 2013 Feb 1. Cell Commun Adhes. 2013. PMID: 23368972 Review.
-
Pemphigus vulgaris: recent advances in our understanding of its pathogenesis.Minerva Stomatol. 2007 Apr;56(4):215-23. Minerva Stomatol. 2007. PMID: 17452959 Review. English, Italian.
Cited by
-
Desmosomal cadherin tension loss in pemphigus vulgaris mediated by the inhibition of active RhoA at cell-cell adhesions.iScience. 2025 Jul 9;28(8):113081. doi: 10.1016/j.isci.2025.113081. eCollection 2025 Aug 15. iScience. 2025. PMID: 40740498 Free PMC article.
-
Data-driven image analysis to determine antibody-induced dissociation of cell-cell adhesion and antibody pathogenicity in pemphigus.PNAS Nexus. 2025 Jul 18;4(8):pgaf218. doi: 10.1093/pnasnexus/pgaf218. eCollection 2025 Aug. PNAS Nexus. 2025. PMID: 40757235 Free PMC article.
-
Autoantibody-Specific Signalling in Pemphigus.Front Med (Lausanne). 2021 Aug 9;8:701809. doi: 10.3389/fmed.2021.701809. eCollection 2021. Front Med (Lausanne). 2021. PMID: 34434944 Free PMC article. Review.
-
Desmosomal Cadherin Tension Loss in Pemphigus Vulgaris Mediated by the Inhibition of Active RhoA at Cell-Cell Adhesions.bioRxiv [Preprint]. 2024 May 6:2024.05.03.592394. doi: 10.1101/2024.05.03.592394. bioRxiv. 2024. Update in: iScience. 2025 Jul 09;28(8):113081. doi: 10.1016/j.isci.2025.113081. PMID: 38766211 Free PMC article. Updated. Preprint.
-
Data-Driven Image Analysis to Determine Antibody-Induced Dissociation of Cell-Cell Adhesion and Antibody Pathogenicity in Pemphigus Vulgaris.bioRxiv [Preprint]. 2024 Oct 13:2024.10.09.617446. doi: 10.1101/2024.10.09.617446. bioRxiv. 2024. Update in: PNAS Nexus. 2025 Jul 18;4(8):pgaf218. doi: 10.1093/pnasnexus/pgaf218. PMID: 39416220 Free PMC article. Updated. Preprint.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

