Gene engineered mesenchymal stem cells: greater transgene expression and efficacy with minicircle vs. plasmid DNA vectors in a mouse model of acute lung injury
- PMID: 33726829
- PMCID: PMC7962085
- DOI: 10.1186/s13287-021-02245-5
Gene engineered mesenchymal stem cells: greater transgene expression and efficacy with minicircle vs. plasmid DNA vectors in a mouse model of acute lung injury
Abstract
Background: Acute lung injury (ALI) and in its severe form, acute respiratory distress syndrome (ARDS), results in increased pulmonary vascular inflammation and permeability and is a major cause of mortality in many critically ill patients. Although cell-based therapies have shown promise in experimental ALI, strategies are needed to enhance the potency of mesenchymal stem cells (MSCs) to develop more effective treatments. Genetic modification of MSCs has been demonstrated to significantly improve the therapeutic benefits of these cells; however, the optimal vector for gene transfer is not clear. Given the acute nature of ARDS, transient transfection is desirable to avoid off-target effects of long-term transgene expression, as well as the potential adverse consequences of genomic integration.
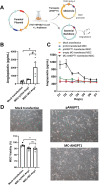
Methods: Here, we explored whether a minicircle DNA (MC) vector containing human angiopoietin 1 (MC-ANGPT1) can provide a more effective platform for gene-enhanced MSC therapy of ALI/ARDS.
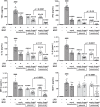
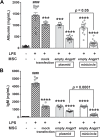
Results: At 24 h after transfection, nuclear-targeted electroporation using an MC-ANGPT1 vector resulted in a 3.7-fold greater increase in human ANGPT1 protein in MSC conditioned media compared to the use of a plasmid ANGPT1 (pANGPT1) vector (2048 ± 567 pg/mL vs. 552.1 ± 33.5 pg/mL). In the lipopolysaccharide (LPS)-induced ALI model, administration of pANGPT1 transfected MSCs significantly reduced bronchoalveolar lavage (BAL) neutrophil counts by 57%, while MC-ANGPT1 transfected MSCs reduced it by 71% (p < 0.001) by Holm-Sidak's multiple comparison test. Moreover, compared to pANGPT1, the MC-ANGPT1 transfected MSCs significantly reduced pulmonary inflammation, as observed in decreased levels of proinflammatory cytokines, such as tumor necrosis factor-alpha (TNF-α), interferon-gamma (IFN-γ), interleukin-6 (IL-6), monocyte chemoattractant protein-1 (MCP-1), and macrophage inflammatory protein-2 (MIP-2). pANGPT1-transfected MSCs significantly reduced BAL albumin levels by 71%, while MC-ANGPT1-transfected MSCs reduced it by 85%.
Conclusions: Overall, using a minicircle vector, we demonstrated an efficient and sustained expression of the ANGPT1 transgene in MSCs and enhanced the therapeutic effect on the ALI model compared to plasmid. These results support the potential benefits of MC-ANGPT1 gene enhancement of MSC therapy to treat ARDS.
Keywords: Acute lung injury; Acute respiratory distress syndrome; Mesenchymal stem cell; Minicircle.
Conflict of interest statement
The funding institution had no role in the conception, design or conduct of the study, data collection or analysis, interpretation or presentation of the data, or preparation, review, or approval of the manuscript. We would also like to declare the following conflicts of interest: DJS holds a patent for MSC therapy for the treatment of acute lung injury and SHJM and YD have received personal fees from Northern Therapeutics Inc. that are outside of this submitted work. The remaining authors have disclosed that they do not have any conflicts of interest.
Figures

Similar articles
-
Prevention of LPS-induced acute lung injury in mice by mesenchymal stem cells overexpressing angiopoietin 1.PLoS Med. 2007 Sep;4(9):e269. doi: 10.1371/journal.pmed.0040269. PLoS Med. 2007. PMID: 17803352 Free PMC article.
-
Human Umbilical Cord Mesenchymal Stem Cells Promote Macrophage PD-L1 Expression and Attenuate Acute Lung Injury in Mice.Curr Stem Cell Res Ther. 2022;17(6):564-575. doi: 10.2174/1574888X17666220127110332. Curr Stem Cell Res Ther. 2022. PMID: 35086457
-
Mesenchymal stem cells overexpressing heme oxygenase-1 ameliorate lipopolysaccharide-induced acute lung injury in rats.J Cell Physiol. 2019 May;234(5):7301-7319. doi: 10.1002/jcp.27488. Epub 2018 Oct 26. J Cell Physiol. 2019. PMID: 30362554
-
Therapeutic implications of mesenchymal stem cells in acute lung injury/acute respiratory distress syndrome.Stem Cell Res Ther. 2013 May 2;4(3):45. doi: 10.1186/scrt193. Stem Cell Res Ther. 2013. PMID: 23673003 Free PMC article. Review.
-
Recent progress in mesenchymal stem cell-based therapy for acute lung injury.Cell Tissue Bank. 2024 Jun;25(2):677-684. doi: 10.1007/s10561-024-10129-0. Epub 2024 Mar 11. Cell Tissue Bank. 2024. PMID: 38466563 Review.
Cited by
-
Advances in acute respiratory distress syndrome: focusing on heterogeneity, pathophysiology, and therapeutic strategies.Signal Transduct Target Ther. 2025 Mar 7;10(1):75. doi: 10.1038/s41392-025-02127-9. Signal Transduct Target Ther. 2025. PMID: 40050633 Free PMC article. Review.
-
Applications of Modified Mesenchymal Stem Cells as Targeted Systems against Tumor Cells.Int J Mol Sci. 2024 Jul 16;25(14):7791. doi: 10.3390/ijms25147791. Int J Mol Sci. 2024. PMID: 39063032 Free PMC article. Review.
-
Mesenchymal Stem Cells as a Gene Delivery Tool: Promise, Problems, and Prospects.Pharmaceutics. 2021 Jun 7;13(6):843. doi: 10.3390/pharmaceutics13060843. Pharmaceutics. 2021. PMID: 34200425 Free PMC article. Review.
-
Innovative nonviral gene delivery strategies for engineering human mesenchymal stem cell phenotypes toward clinical applications.Curr Opin Biotechnol. 2022 Dec;78:102819. doi: 10.1016/j.copbio.2022.102819. Epub 2022 Oct 21. Curr Opin Biotechnol. 2022. PMID: 36274497 Free PMC article. Review.
-
Immunomodulatory Mechanisms of Mesenchymal Stem Cells and Their Potential Clinical Applications.Int J Mol Sci. 2022 Sep 2;23(17):10023. doi: 10.3390/ijms231710023. Int J Mol Sci. 2022. PMID: 36077421 Free PMC article. Review.
References
-
- Thompson BT, Chambers RC, Liu KD. Acute respiratory distress syndrome. N Engl J Med. 2017;377(19):1904–1905. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

