Bardet-Biedl syndrome-7 (BBS7) shows treatment potential and a cone-rod dystrophy phenotype that recapitulates the non-human primate model
- PMID: 33729075
- PMCID: PMC8743897
- DOI: 10.1080/13816810.2021.1888132
Bardet-Biedl syndrome-7 (BBS7) shows treatment potential and a cone-rod dystrophy phenotype that recapitulates the non-human primate model
Abstract
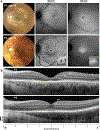
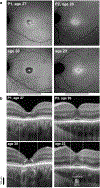
Purpose: To provide a detailed ophthalmic phenotype of two male patients with Bardet-Biedl Syndrome (BBS) due to mutations in the BBS7 geneMethods: Two brothers ages 26 (Patient 1, P1) and 23 (P2) underwent comprehensive ophthalmic evaluations over three years. Visual function was assessed with full-field electroretinograms (ffERGs), kinetic and chromatic perimetry, multimodal imaging with spectral domain optical coherence tomography (SD-OCT), fundus autofluorescence (FAF) with short- (SW) and near-infrared (NIR) excitation lights and adaptive optics scanning light ophthalmoscopy (AOSLO).Results: Both siblings had a history of obesity and postaxial polydactyly; P2 had diagnoses of type 1 Diabetes Mellitus, Addison's disease, high-functioning autism-spectrum disorder and -12D myopia. Visual acuities were better than 20/30. Kinetic fields were moderately constricted. Cone-mediated ffERGs were undetectable, rod ERGs were ~80% of normal mean. Static perimetry showed severe central cone and rod dysfunction. Foveal to parafoveal hypoautofluorescence, most obvious on NIR-FAF, co-localized with outer segment shortening/loss and outer nuclear layer thinning by SD-OCT, and with reduced photoreceptors densities by AOSLO. A structural-functional dissociation was confirmed for cone- and rod-mediated parameters. Worsening of the above abnormalities was documented by SD-OCT and FAF in P2 at 3 years. Gene screening identified compound heterozygous mutations in BBS7 (p.Val266Glu: c.797 T > A of maternal origin; c.1781_1783delCAT, paternal) in both patients.Conclusions: BBS7-associated retinal degeneration may present as a progressive cone-rod dystrophy pattern, reminiscent of both the murine and non-human primate models of the disease. Predominantly central retinal abnormalities in both cone and rod photoreceptors showed a structural-functional dissociation, an ideal scenario for gene augmentation treatments.
Keywords: BBS; BBS7; Bardet-Biedl; OCT; RP; adaptive optics ophthalmoscopy; cone dystrophy; cone-rod dystrophy; retinitis pigmentosa.
Conflict of interest statement
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of this article.
Figures




References
-
- Green JS, Parfrey PS, Harnett JD, Farid NR, Cramer BC, Johnson G, Heath O, McManamon PJ, O’Leary E, Pryse-Phillips W, et al. The cardinal manifestations of Bardet-Biedl syndrome, a form of Laurence-Moon-Biedl syndrome. N Engl J Med. 1989;321:1002–09. - PubMed
-
- Forsyth RL, Gunay-Aygun M. Bardet-Biedl Syndrome Overview. 2003. July 14 [Updated 2020 Jul 23]. In: Adam MP, Ardinger HH, Pagon RA, et al. , editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2021. Available from: https://www-ncbi-nlm-nih-gov.proxy.library.upenn.edu/books/NBK1363/.
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
