The pharmacokinetics and drug-drug interactions of ivermectin in Aedes aegypti mosquitoes
- PMID: 33730100
- PMCID: PMC7968666
- DOI: 10.1371/journal.ppat.1009382
The pharmacokinetics and drug-drug interactions of ivermectin in Aedes aegypti mosquitoes
Abstract
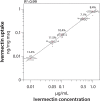
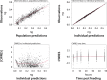
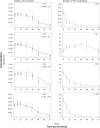
Mosquitoes are vectors of major diseases such as dengue fever and malaria. Mass drug administration of endectocides to humans and livestock is a promising complementary approach to current insecticide-based vector control measures. The aim of this study was to establish an insect model for pharmacokinetic and drug-drug interaction studies to develop sustainable endectocides for vector control. Female Aedes aegypti mosquitoes were fed with human blood containing either ivermectin alone or ivermectin in combination with ketoconazole, rifampicin, ritonavir, or piperonyl butoxide. Drug concentrations were quantified by LC-MS/MS at selected time points post-feeding. Primary pharmacokinetic parameters and extent of drug-drug interactions were calculated by pharmacometric modelling. Lastly, the drug effect of the treatments was examined. The mosquitoes could be dosed with a high precision (%CV: ≤13.4%) over a range of 0.01-1 μg/ml ivermectin without showing saturation (R2: 0.99). The kinetics of ivermectin were characterised by an initial lag phase of 18.5 h (CI90%: 17.0-19.8 h) followed by a slow zero-order elimination rate of 5.5 pg/h (CI90%: 5.1-5.9 pg/h). By contrast, ketoconazole, ritonavir, and piperonyl butoxide were immediately excreted following first order elimination, whereas rifampicin accumulated over days in the mosquitoes. Ritonavir increased the lag phase of ivermectin by 11.4 h (CI90%: 8.7-14.2 h) resulting in an increased exposure (+29%) and an enhanced mosquitocidal effect. In summary, this study shows that the pharmacokinetics of drugs can be investigated and modulated in an Ae. aegypti animal model. This may help in the development of novel vector-control interventions and further our understanding of toxicology in arthropods.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
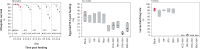
References
-
- World health organization (2020). Vector-borne diseases, Fact sheet no. 387 2014. Available from: https://www.who.int/news-room/fact-sheets/detail/vector-borne-diseases. [Accessed: 03-February-2021].
-
- World health organization (2019). World malaria report 2019: Geneva. Available from https://www.who.int/publications/i/item/world-malaria-report-2019. [Accessed: 03-February-2021].
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

