Natural Polyphyllins (I, II, D, VI, VII) Reverses Cancer Through Apoptosis, Autophagy, Mitophagy, Inflammation, and Necroptosis
- PMID: 33732000
- PMCID: PMC7956893
- DOI: 10.2147/OTT.S287354
Natural Polyphyllins (I, II, D, VI, VII) Reverses Cancer Through Apoptosis, Autophagy, Mitophagy, Inflammation, and Necroptosis
Abstract
Cancer is the second leading cause of mortality worldwide. Conventional therapies, including surgery, radiation, and chemotherapy, have limited success because of secondary resistance. Therefore, safe, non-resistant, less toxic, and convenient drugs are urgently required. Natural products (NPs), primarily sourced from medicinal plants, are ideal for cancer treatment because of their low toxicity and high success. NPs cure cancer by regulating different pathways, such as PI3K/AKT/mTOR, ER stress, JNK, Wnt, STAT3, MAPKs, NF-kB, MEK-ERK, inflammation, oxidative stress, apoptosis, autophagy, mitophagy, and necroptosis. Among the NPs, steroid saponins, including polyphyllins (I, II, D, VI, and VII), have potent pharmacological, analgesic, and anticancer activities for the induction of cytotoxicity. Recent research has demonstrated that polyphyllins (PPs) possess potent effects against different cancers through apoptosis, autophagy, inflammation, and necroptosis. This review summarizes the available studies on PPs against cancer to provide a basis for future research.
Keywords: apoptosis; autophagy; inflammation; natural products; necroptosis; polyphyllins; saponins.
© 2021 Ahmad et al.
Conflict of interest statement
All authors declare that they have no competing interests in this work.
Figures

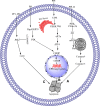
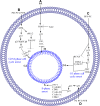
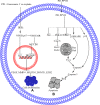
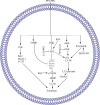
References
-
- Pengyu SAB, Lijuan Z. Natural β-elemene: advances in targeting cancer through different molecular pathways. North Am J Aced Res. 2018;1(4):27.
-
- Su P, Ahmad B, Ahmad M, et al. Targeting Cancer through PI3K/AKT/mTOR pathway with selected natural products (β-Elemene, Puerarin and Gypenosides). Am J Biomed Sci Res. 2020;8:4.
-
- Pengyu SBA, Ullah H, Hussain B, Khan SZ, Mohammad SAJK, Ahmad M. Targeting cancer through autophagy with Β-Elemene and Puerarin. Am J Biomed Sci Res. 2020;8.
-
- Organization WH. Global Health Observatory Data Repository. 2011. Number of Deaths (World) by Cause; 2015.
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

